When Does Water Freeze: Key Insights Unveiled
Water freezing is a phenomenon many have observed but few understand in its full scientific depth. Knowing when water freezes isn’t just academic; it has practical implications, from ensuring pipes don’t burst in winter to managing food preservation strategies. This article delves into the intricate science behind water’s freezing point and offers expert insights to help you grasp this essential concept.
Key Insights
- Water freezes at 0°C (32°F) under standard atmospheric pressure.
- Altitude and impurities can alter this freezing point.
- Understanding these factors can help prevent property damage during cold spells.
The Science Behind Water Freezing
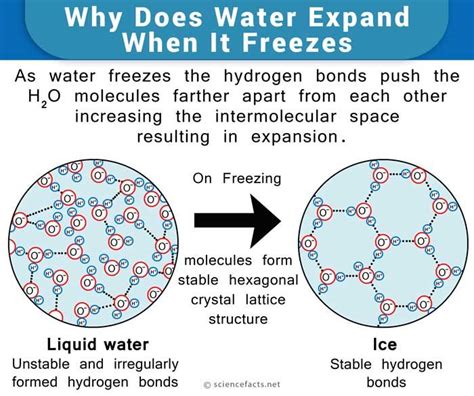
Water freezes at its freezing point, which is 0°C (32°F) under standard atmospheric pressure of 1 atmosphere. This is when water molecules slow down and arrange into a crystalline structure, forming ice. The freezing point of water is determined by several factors, including its chemical composition and environmental conditions.
The molecular structure of water (H2O) plays a crucial role in its freezing behavior. Each water molecule consists of two hydrogen atoms bonded to a single oxygen atom, forming a bent shape. When water cools, these molecules move closer together and form hydrogen bonds. However, as they reach 0°C, these bonds are strong enough to create a lattice structure, leading to the solid state of ice.
Impact of Altitude and Impurities
While 0°C is the freezing point at sea level under standard conditions, altitude and impurities can significantly alter this freezing point. At higher altitudes, where atmospheric pressure is lower, water can freeze at temperatures slightly below 0°C. Conversely, impurities such as salt, sugar, or other solutes can depress the freezing point—a phenomenon known as freezing point depression.
For example, seawater, which contains a high concentration of salts, freezes at a lower temperature than pure water. This is why coastal areas often experience milder winters despite lower temperatures, because the salt content in seawater lowers its freezing point. This principle is also used in antifreeze solutions, where the addition of ethylene glycol prevents water from freezing at typical winter temperatures.
Applications and Implications
Understanding when water freezes has numerous practical applications. In residential settings, knowing that water will freeze at 0°C helps in taking preventive measures against frozen pipes during cold weather. Homeowners can insulate pipes and maintain indoor temperatures above 32°F to prevent freezing and subsequent burst pipes. Additionally, in food storage, keeping refrigerator and freezer temperatures below 0°C ensures that water-based substances remain in solid form, preserving the texture and quality of perishable items.
In industrial processes, freezing points are critical in applications like cooling systems for machinery, where maintaining a steady temperature is necessary for optimal operation. Similarly, in the food industry, understanding the freezing point of water helps in preserving foods, making ice, and controlling the texture of frozen products.
What happens to water at higher altitudes?
At higher altitudes, the atmospheric pressure decreases. This causes water to freeze at temperatures slightly below 0°C, because the reduced pressure lowers the freezing point.
Can impurities prevent water from freezing?
Yes, impurities like salt, sugar, or other solutes can depress the freezing point of water, making it freeze at lower temperatures. This principle is used in antifreeze solutions to prevent vehicle coolants from freezing.
In conclusion, understanding when water freezes requires a grasp of its molecular behavior and environmental influences. With practical implications ranging from home maintenance to industrial processes, knowledge of this scientific principle is invaluable. Ensuring you have the right strategies in place can save you from property damage, maintain the quality of your food, and ensure efficient operation of machinery.