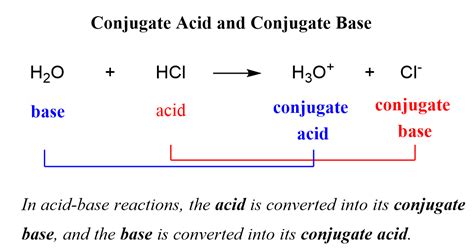
Understanding the concept of a conjugate base is fundamental to grasping the intricacies of acid-base chemistry. In essence, a conjugate base is formed when an acid donates a proton (H⁺) in a reaction, resulting in the base’s production. This concept is pivotal in both theoretical and practical chemistry, particularly in fields such as biochemistry, pharmaceuticals, and environmental science. In this guide, we delve into what a conjugate base is, its applications, and real-life examples.
Key Insights
- Primary insight with practical relevance: Recognizing conjugate bases helps in understanding reaction mechanisms and predicting the behavior of substances in various environments.
- Technical consideration with clear application: In a neutralization reaction, the conjugate base of an acid interacts with the conjugate acid of a base, making it a central concept in buffer solutions.
- Actionable recommendation: For chemistry students and professionals, mastering the relationship between acids, bases, and their conjugates is crucial for advanced studies and practical applications.
Acid-Base Interactions and Conjugate Bases
To fully appreciate the role of a conjugate base, one must first understand the nature of acids and bases. An acid is a substance that donates protons in a chemical reaction, whereas a base accepts these protons. The conjugate base of an acid is the molecule that remains after the acid has donated a proton. For instance, when hydrochloric acid (HCl) donates a proton, it forms water (H₂O), which acts as the conjugate base. This interaction illustrates the direct relationship between acids and their conjugate bases.
Conjugate Bases in Buffer Solutions
Buffer solutions are critical in maintaining the pH stability of a solution. They comprise a weak acid and its conjugate base, or a weak base and its conjugate acid. The ability of buffers to resist pH changes is a direct result of the presence of these conjugate pairs. Consider the bicarbonate buffer system in human blood. Carbonic acid (H₂CO₃) and its conjugate base, bicarbonate (HCO₃⁻), work together to maintain blood pH within a narrow range, demonstrating the practical importance of conjugate bases in biological systems.
Another example includes the acetate buffer system, which is widely used in laboratories. Acetic acid (CH₃COOH) acts as the acid, and its conjugate base, acetate (CH₃COO⁻), works to maintain a stable pH. The efficacy of these buffers in laboratory settings underscores the importance of conjugate bases in scientific research.
What is an example of a conjugate base?
A classic example is the conjugate base of acetic acid (CH₃COOH), which is acetate (CH₃COO⁻). When acetic acid donates a proton, it forms water and leaves behind acetate as its conjugate base.
Why is it important to study conjugate bases?
Understanding conjugate bases is crucial for predicting reaction outcomes in various chemical environments, aiding in the design of effective buffer solutions, and comprehending the behavior of acids and bases in biochemical processes.
In conclusion, the concept of a conjugate base plays a vital role in both the theoretical and practical realms of chemistry. By recognizing how acids donate protons and form their respective conjugate bases, one can better predict chemical behavior and leverage this knowledge in diverse applications, from biological systems to laboratory experiments.