Water is a fundamental element that exists in three states: solid (ice), liquid (water), and gas (water vapor). Understanding the exact temperature at which water freezes is crucial in various contexts, ranging from weather prediction to everyday life. This guide will equip you with comprehensive knowledge and practical steps to determine the exact degree Fahrenheit for freezing water, addressing common user concerns and providing actionable advice.
Understanding the freezing point of water helps in many practical applications. For example, if you’re a traveler, knowing the precise temperature can help you predict and prepare for cold weather. For those in the food industry, it’s important to ensure that freezing conditions are met to preserve food quality. This guide provides clear, step-by-step advice, backed by real-world examples to ensure you can easily apply this knowledge to solve common problems.
Quick Reference
Quick Reference
- Immediate action item: Check that your thermometer is calibrated correctly to get the exact freezing point.
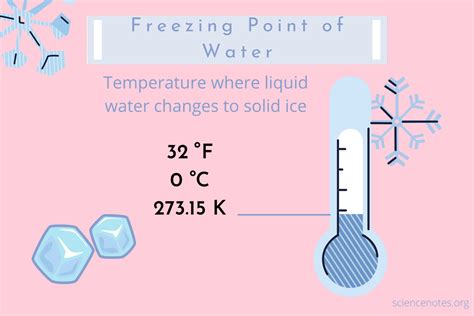
- Essential tip: The freezing point of pure water is 32°F (0°C). Always use a calibrated thermometer for accuracy.
- Common mistake to avoid: Assuming seawater freezes at the same temperature as freshwater; seawater freezes at a lower temperature due to dissolved salts.
Whether you’re dealing with an outdoor project or scientific experiment, understanding the freezing point is vital. Here are detailed steps and practical examples to ensure you can accurately determine when water freezes in Fahrenheit.
Understanding the Freezing Point of Water
The freezing point of water is a fundamental concept in both science and everyday life. Pure water freezes at exactly 32°F (0°C) under standard atmospheric pressure. This information is critical for numerous applications. Here’s a step-by-step guide to ensure you understand and can apply this knowledge accurately.
- Define Standard Atmospheric Pressure: The freezing point of water is defined at standard atmospheric pressure, which is 1 atmosphere (atm) or approximately 101.3 kPa. This reference helps to standardize the measurement across different environments.
- Understand the Composition of Water: Pure water consists of H2O, where hydrogen and oxygen atoms form a stable molecule. Impurities such as minerals and salts can lower the freezing point, a concept known as freezing point depression.
- Check Thermometer Calibration: Ensure that your thermometer is properly calibrated. Different thermometers, whether digital or analog, can vary slightly in readings. Calibrate against known reference points.
- Measure Temperature in Fahrenheit: When measuring the freezing point in Fahrenheit, start by placing the thermometer in a container of water. Slowly lower the temperature until the water begins to freeze. This gradual process helps avoid misreadings.
- Observe the Freezing Point: Look for signs of freezing, such as the formation of ice crystals on the surface of the water or the temperature stabilizing at 32°F. These indicators confirm the exact freezing point.
By following these steps, you’ll be able to accurately determine the freezing point of water in Fahrenheit, ensuring precise measurements for both scientific and practical applications.
Practical Applications of Knowing the Freezing Point
Understanding the exact degree Fahrenheit for freezing water opens a world of practical applications. Here’s a detailed breakdown of various scenarios where this knowledge comes into play, along with actionable tips and real-world examples.
Scientific Experiments
In scientific research, knowing the exact freezing point of water is essential for controlled experiments. For instance, in environmental science, researchers might need to simulate freezing conditions to study the effects on ecosystems or materials. Here’s how you can apply this knowledge:
- Controlled Experiments: When conducting experiments that require specific temperature conditions, ensure that your setup can maintain a consistent temperature. Use precise thermometers and thermostatic devices to monitor and control the freezing point accurately.
- Data Collection: Record temperature readings at the moment water begins to freeze. This data can be used to compare with other variables, such as the presence of impurities or the type of container used.
- Analyze Results: After experiments, analyze how deviations from the standard freezing point (32°F) affect outcomes. This analysis can provide valuable insights into factors influencing freezing points.
Food Preservation
For those involved in food preservation, accurately knowing the freezing point ensures that food is stored at the right temperatures to prevent spoilage. Here’s how you can apply this knowledge:
- Freezing Food: Store perishable items at or below 32°F to ensure they freeze quickly and maintain quality. Use freezers that are calibrated to precise temperatures.
- Shelf Life: Proper freezing extends the shelf life of food. By freezing at the exact temperature, you reduce the risk of bacterial growth and freezer burn.
- Defrosting: When defrosting frozen items, monitor the thawing process carefully to prevent temperature spikes that could affect food quality.
Weather and Environmental Science
In weather forecasting and environmental studies, knowing the freezing point helps predict and prepare for cold weather conditions. Here’s how this knowledge can be applied:
- Weather Predictions: Meteorologists use the freezing point of water to predict frost, ice formation, and other cold-related weather phenomena. Understanding this helps in issuing accurate warnings.
- Hydrological Studies: For studies involving water bodies, knowing the freezing point helps in understanding ice formation, seasonal changes, and the impact of global warming on frozen regions.
- Urban Planning: City planners use this knowledge to design infrastructure that can withstand freezing temperatures, such as roads, bridges, and utilities.
Practical FAQ
Why is the freezing point of water important?
The freezing point of water is important for various reasons including scientific research, food preservation, weather forecasting, and urban planning. It helps in controlling experimental conditions, ensuring food safety, predicting weather conditions, and designing infrastructure that can withstand cold temperatures.
How do impurities affect the freezing point of water?
Impurities such as salts, minerals, and other dissolved substances lower the freezing point of water. This phenomenon is known as freezing point depression. For example, seawater freezes at a lower temperature than freshwater due to the presence of salts.
What should I do if my thermometer doesn’t read 32°F when water freezes?
If your thermometer does not read 32°F when water freezes, it may need calibration. First, ensure the thermometer is placed in a large, stable container of water. Use multiple thermometers for cross-verification. If discrepancies persist, calibrate the thermometer using standard reference points available at your local meteorological office or scientific institution.
By understanding the exact degree Fahrenheit for freezing water, you can address various practical challenges efficiently. This guide has provided a detailed, step-by-step approach to determining and applying this crucial temperature, ensuring you can rely on accurate measurements in both scientific and everyday contexts.
Remember, precise knowledge of the freezing point of water at 32°F is not just a scientific fact; it’s a practical tool that helps in various fields, ensuring safety, efficiency, and accuracy. Use this guide to enhance your understanding and apply it in real-world situations.