Understanding the intricate world of electrochemical cells can often feel like navigating a maze, especially when you come across terms like “voltaic cell” and “electrolytic cell.” Both types of cells are pivotal in various applications ranging from powering your smartphone to enabling advanced research in biochemistry. This guide will illuminate the key differences between these two fascinating cell types, offering step-by-step guidance with actionable advice, real-world examples, and practical solutions to address common user pain points.
Whether you’re a student grappling with electrochemical principles or an engineer exploring applications, this guide will help demystify the concepts and clarify the practical differences. We’ll start with a detailed problem-solution opening to set the stage, followed by a quick reference guide packed with essential insights, a deep dive into the specific functionalities of both types of cells, and frequently asked questions. Ready? Let’s embark on this enlightening journey.
Addressing Your Need for Clarity
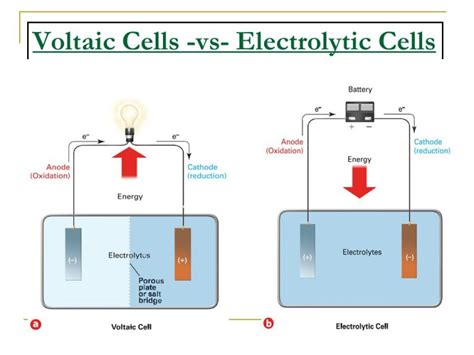
At its core, the confusion between voltaic and electrolytic cells often boils down to their distinct functions and applications. A voltaic cell generates electrical energy from spontaneous redox reactions, which is useful for applications like batteries and fuel cells. In contrast, an electrolytic cell uses electrical energy to drive non-spontaneous redox reactions, which is crucial for processes such as electroplating and industrial chemical production. By understanding these fundamental differences, you’ll be able to choose the right type of cell for your specific needs. This guide will break down each cell’s role, offering practical tips and examples to ensure you’re fully equipped to make informed decisions.
Quick Reference
Quick Reference
- Immediate action item: Identify if you need a cell to generate electrical energy or drive a chemical reaction.
- Essential tip: Use a voltaic cell for spontaneous reactions (e.g., battery operation), and an electrolytic cell for non-spontaneous reactions (e.g., electrolysis).
- Common mistake to avoid: Confusing the direction of electron flow and reaction spontaneity. Ensure you understand whether the process is exothermic (releases energy) or endothermic (requires energy).
The Voltaic Cell: Generating Energy Through Spontaneous Reactions
Voltaic cells, also known as galvanic cells, operate through spontaneous redox reactions. Essentially, they convert chemical energy into electrical energy through a redox reaction. Here’s how they work:
A voltaic cell typically consists of two different metals (or a metal and a non-metal) placed in separate compartments containing electrolyte solutions. When connected, a redox reaction occurs, with electrons flowing from one metal to the other. This flow of electrons generates an electric current.
How it Works
Let’s delve into a practical example: the classic zinc-copper cell.
1. Anode: The zinc strip serves as the anode. Zinc spontaneously oxidizes, releasing electrons. The reaction is:
Zn(s) → Zn²⁺(aq) + 2e⁻
2. Cathode: The copper strip serves as the cathode. The released electrons from the zinc are accepted by copper ions, reducing them to copper metal. The reaction is:
Cu²⁺(aq) + 2e⁻ → Cu(s)
3. Electrolyte: Salt bridges or permeable membranes connect the two half-cells, allowing ions to migrate and maintain electrical neutrality.
4. Electrochemical Circuit: The entire system forms an electrochemical circuit. The electrons flow from the zinc anode to the copper cathode through an external circuit, generating electrical energy.
Voltaic cells are ubiquitous in everyday applications. Consider the AA batteries in your remote control or the lead-acid batteries in your car. These cells convert chemical energy from the redox reactions into electrical energy, powering everything from simple toys to complex machinery.
Practical Tips and Best Practices
Here are some essential tips and best practices for working with voltaic cells:
- Ensure that your redox reactions are spontaneous. Non-spontaneous reactions won’t drive the desired current.
- Use different metals with different standard reduction potentials to maximize the voltage output of your cell.
- Always keep your cell components clean and dry to avoid unwanted side reactions.
Remember, the goal is to harness the natural energy from a spontaneous reaction to produce a reliable electric current.
The Electrolytic Cell: Driving Reactions Through External Power
Electrolytic cells, on the other hand, use electrical energy to drive non-spontaneous redox reactions. These cells are crucial for applications that require the synthesis of specific chemicals that aren’t naturally occurring. Here’s how they function:
An electrolytic cell typically consists of two electrodes submerged in an electrolyte solution. An external power source is used to apply voltage across the electrodes, forcing an otherwise non-spontaneous redox reaction to occur.
How it Works
Consider an electrolytic cell designed for electrolysis of water:
1. Anode: At the anode, water molecules lose electrons and oxidize to produce oxygen gas. The reaction is:
2H₂O(l) → O₂(g) + 4H⁺(aq) + 4e⁻
2. Cathode: At the cathode, hydrogen ions gain electrons and reduce to produce hydrogen gas. The reaction is:
4H⁺(aq) + 4e⁻ → 2H₂(g)
3. Electrolyte: An electrolyte solution (e.g., sulfuric acid) enhances the flow of ions between the electrodes.
4. Power Source: An external power source provides the necessary voltage to drive the non-spontaneous reaction, separating water into hydrogen and oxygen gases.
Electrolytic cells are invaluable in industrial processes like the purification of metals and the production of chemicals like chlorine and sodium hydroxide. They’re also used in everyday applications such as electroplating to coat objects with a thin layer of metal.
Practical Tips and Best Practices
Here are some practical tips and best practices for working with electrolytic cells:
- Ensure the applied voltage is sufficient to drive the desired reaction. Too little voltage won’t achieve the desired result, while too much can cause unwanted side reactions.
- Use inert electrodes (like platinum) to avoid unwanted reactions at the electrode surface.
- Always use high-quality electrolytes to ensure efficient ion transport and reaction rates.
Electrolytic cells harness external electrical energy to push chemical reactions in a controlled manner, enabling the synthesis of materials and chemicals that would otherwise be difficult to obtain.
Frequently Asked Questions
What is the main difference between a voltaic and electrolytic cell?
The main difference lies in the direction of energy flow and the type of reactions they drive. A voltaic cell generates electrical energy from spontaneous redox reactions, while an electrolytic cell uses electrical energy to drive non-spontaneous redox reactions. In simple terms, voltaic cells convert chemical energy into electrical energy, and electrolytic cells use electrical energy to drive chemical reactions.
Can a voltaic cell be used for electrolysis?
No, a voltaic cell cannot be used for electrolysis because it relies on spontaneous redox reactions to generate electrical energy. Electrolysis requires an external power source to drive non-spontaneous reactions, which a voltaic cell does not provide. For electrolysis, you need an electrolytic cell connected to an external power source.
What materials are commonly used in voltaic and electrolytic cells?
Voltaic cells often use metals with distinct electrochemical potentials (e.