When embarking on scientific research or educational projects that require a thorough understanding of thermal energy, the thermal energy formula is an essential component you’ll frequently encounter. However, grasping the intricacies of this formula can often be daunting. This guide aims to demystify thermal energy calculations with clear, actionable advice, practical examples, and easy-to-follow instructions. By the end of this guide, you will have a robust understanding of how to apply the thermal energy formula in real-world scenarios, addressing both basic and advanced levels of user needs.
Problem-Solution Opening Addressing User Needs
Understanding and calculating thermal energy is crucial for various scientific fields, including physics, chemistry, and engineering. The thermal energy formula is a fundamental concept, often appearing in experiments and theoretical studies. However, it can be challenging to grasp, especially for those new to these fields. Many users encounter difficulties in applying this formula due to a lack of practical, hands-on guidance. This guide addresses these pain points by offering step-by-step guidance, real-world examples, and actionable tips to ensure you can master this formula with confidence. Whether you are conducting a simple experiment or diving into complex thermal dynamics, this guide will equip you with the knowledge and tools you need to succeed.
Quick Reference
- Immediate action item with clear benefit: Calculate initial thermal energy using Q = mcΔT to determine the energy change in a substance.
- Essential tip with step-by-step guidance: Start by identifying the mass, specific heat capacity, and temperature change, then plug these into the formula.
- Common mistake to avoid with solution: Confusing specific heat capacity with general heat; ensure you use the substance-specific value.
Mastering the Basics: Understanding the Thermal Energy Formula
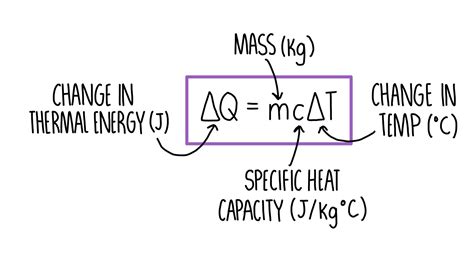
To start with the thermal energy formula, you need to understand its components and significance. The formula for thermal energy is given by:
Q = mcΔT
Where:
Q = thermal energy (Joules, J)
m = mass of the substance (kg)
c = specific heat capacity of the substance (J/kg·K)
ΔT = change in temperature (K or °C)
The thermal energy formula allows you to calculate the amount of heat required to change the temperature of a given mass of a substance.
Let’s break down a practical example to make this formula more understandable.
Imagine you’re tasked with heating a 2-kilogram block of aluminum from 20°C to 80°C. Here’s how you would apply the formula:
- Identify the variables:
- m = 2 kg
- c = 900 J/kg·K (specific heat capacity of aluminum)
- Initial temperature (T_i) = 20°C
- Final temperature (T_f) = 80°C
- Calculate the change in temperature:
- Insert values into the thermal energy formula:
ΔT = T_f - T_i = 80°C - 20°C = 60°C
Q = mcΔT
Q = 2 kg × 900 J/kg·K × 60 K
Q = 108,000 J
This example demonstrates that 108,000 Joules of thermal energy are required to heat the aluminum block from 20°C to 80°C. This practical approach will help solidify your understanding of the formula.
Intermediate Applications: Beyond Basic Calculations
As you become more comfortable with the thermal energy formula, you may encounter more complex scenarios that require its application.
Mixing Two Substances
When mixing two substances and determining the final temperature, you will need to use the thermal energy formula in conjunction with energy conservation principles. Here’s how to approach it:
Suppose you mix 2 kg of water at 20°C with 1 kg of iron at 100°C, and you want to find the final temperature. Follow these steps:
- Calculate the thermal energy lost by the iron:
- Calculate the thermal energy gained by the water:
- Set the heat lost by iron equal to the heat gained by water:
- Solve for T_f:
Q_iron = mcΔT
ΔT_iron = T_i_iron - T_f
Q_iron = 1 kg × 450 J/kg·K × (100°C - T_f)
Q_water = mcΔT
Q_water = 2 kg × 4186 J/kg·K × (T_f - 20°C)
1 kg × 450 J/kg·K × (100°C - T_f) = 2 kg × 4186 J/kg·K × (T_f - 20°C)
450 (100 - T_f) = 8372 (T_f - 20)
45000 - 450T_f = 8372T_f - 167440
45000 + 167440 = 8822T_f
212440 = 8822T_f
T_f = 24.1°C
Thus, the final temperature when mixing 2 kg of water at 20°C with 1 kg of iron at 100°C is 24.1°C.
Advanced Applications: Complex Thermal Energy Problems
For those delving into advanced thermal energy problems, understanding phase changes and chemical reactions is essential. These scenarios require the thermal energy formula to be combined with phase change equations and reaction heats.
Phase Change Calculations
When dealing with phase changes, such as melting or boiling, the latent heat of fusion or vaporization is introduced. The formula changes to:
Q = mL
Where:
L = latent heat of fusion (for melting) or latent heat of vaporization (for boiling) (J/kg)
Let’s consider an example of melting ice:
- Identify the variables:
- m = 1 kg
- L = 334,000 J/kg (latent heat of fusion for ice)
- Calculate the thermal energy required:
Q = mL
Q = 1 kg × 334,000 J/kg
Q = 334,000 J
This calculation shows that 334,000 Joules of thermal energy are required to melt 1 kg of ice at 0°C.
Chemical Reaction Heats
In chemical reactions, the heat involved can be calculated using the reaction’s enthalpy change (ΔH). Here’s a step-by-step approach:
- Determine the enthalpy change:
For the reaction of 2 moles of hydrogen gas with oxygen to form water, the ΔH is -572 kJ/mol.