When delving into the world of chemistry, one inevitably encounters the balancing act of strong acids and bases, two classes of compounds that play a pivotal role in various chemical reactions and processes. Understanding the interplay between these substances is crucial not just for academic purposes but for practical applications ranging from industrial processes to household chores. This guide aims to demystify the concepts behind strong acids and bases, offering you actionable advice and practical solutions to master this aspect of chemistry.
The Challenge: Why Strong Acids and Bases Matter
Strong acids and bases are fundamental to chemistry because they fully dissociate in water, leading to a high concentration of hydrogen (H⁺) or hydroxide (OH⁻) ions. This property makes them incredibly potent in chemical reactions, influencing everything from pH levels in biological systems to industrial manufacturing processes. Despite their power, handling strong acids and bases requires caution due to their potential to cause severe chemical burns and other hazardous effects.
Many users struggle with the correct usage and handling of these substances, leading to mishaps ranging from ineffective reactions to dangerous spills. This guide is designed to address these common pain points by providing step-by-step guidance, real-world examples, and practical solutions to ensure you can manage strong acids and bases effectively and safely.
Quick Reference Guide
Quick Reference
- Immediate action item with clear benefit: Always neutralize spills of strong acids or bases immediately with appropriate counter-substances like a weak base or acid, respectively.
- Essential tip with step-by-step guidance: To dilute a strong acid safely, always add acid to water, not water to acid, to prevent exothermic reactions from causing splattering.
- Common mistake to avoid with solution: Never mix different types of acids or bases as this can lead to unpredictable and hazardous chemical reactions.
Understanding Strong Acids: Dissecting Their Properties and Use
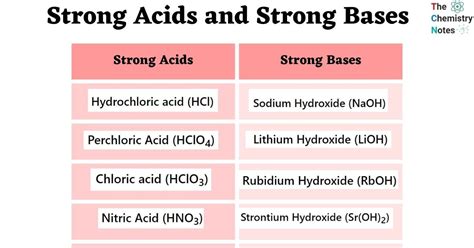
Strong acids are compounds that completely dissociate in aqueous solutions, yielding a high concentration of hydrogen ions (H⁺). Common examples include hydrochloric acid (HCl), sulfuric acid (H₂SO₄), and nitric acid (HNO₃). Recognizing these acids and understanding their behavior is crucial for their correct use in laboratories and industries.
Here’s how to effectively work with strong acids:
- Identification: Learn to identify common strong acids based on their chemical formula and properties.
- Handling and Safety: Always use appropriate personal protective equipment (PPE) such as gloves, goggles, and lab coats when handling strong acids. Avoid contact with skin and eyes, and work in well-ventilated areas or fume hoods.
- Dilution: To dilute a strong acid, always add the acid slowly to water while stirring. This prevents exothermic reactions that could cause the mixture to splatter. The general formula for safe dilution is:
- Neutralization: To neutralize a strong acid spill, use a weak base such as sodium bicarbonate (NaHCO₃). Always apply the neutralizing agent from a distance to avoid reactions that might cause splashing.
| Starting volume of acid (V1) | Concentration of acid (C1) | Desired volume of diluted solution (V2) | Desired concentration (C2) |
|---|---|---|---|
| V1 | C1 | V2 | C2 |
| V1 x C1 = V2 x C2 |
Mastering Strong Bases: Their Characteristics and Applications
Strong bases, also known as strong bases, completely dissociate in water, resulting in high hydroxide ion (OH⁻) concentrations. Examples include sodium hydroxide (NaOH), potassium hydroxide (KOH), and calcium hydroxide (Ca(OH)₂). Understanding the behavior of strong bases is essential for their correct application in various chemical processes.
Here’s a detailed guide on working with strong bases:
- Identification: Familiarize yourself with the chemical formulas and properties of common strong bases.
- Handling and Safety: Use appropriate PPE like gloves and goggles to prevent contact with skin and eyes. Store bases in tightly sealed containers away from moisture and acids.
- Dilution: Dilute strong bases by adding water to the base slowly, stirring constantly. The process is less exothermic than acid dilution but still requires careful handling. The dilution principle remains similar to acids:
- Neutralization: In case of a strong base spill, neutralize using a weak acid like vinegar (acetic acid). Apply the neutralizing agent cautiously to avoid splashes and reactions.
| Starting volume of base (V1) | Concentration of base (C1) | Desired volume of diluted solution (V2) | Desired concentration (C2) |
|---|---|---|---|
| V1 | C1 | V2 | C2 |
| V1 x C1 = V2 x C2 |
Practical FAQ: Answers to Common User Concerns
What are the safety precautions I should follow when handling strong acids and bases?
Safety should always be your top priority when working with strong acids and bases. Here’s a comprehensive list of precautions:
- Wear appropriate PPE including gloves, goggles, and lab coats.
- Avoid direct contact with skin and eyes. In case of contact, immediately rinse the affected area with plenty of water and seek medical attention.
- Work in well-ventilated areas or use fume hoods to avoid inhalation of fumes.
- Store acids and bases in tightly sealed containers away from incompatible substances like water and other acids/bases.
- Label all containers clearly to avoid mix-ups and follow the appropriate disposal procedures for chemical waste.
Advanced Tips for Professional Use
For those seeking to delve deeper into the professional use of strong acids and bases, here are some advanced tips:
- Optimizing Reactions: When conducting chemical reactions involving strong acids or bases, optimize reaction conditions such as temperature, pH, and concentration to ensure efficient and safe outcomes.
- Automated Systems: In industrial settings, consider using automated dispensing and mixing systems that handle acids and bases under controlled conditions, minimizing human contact and reducing risks.
- Regular Training: Engage in regular safety and handling training to stay updated with the latest safety protocols and technological advancements in chemical handling.
- Emergency Response: Develop and practice an emergency response plan for accidental spills or exposures involving strong acids and bases, including the use of eyewash stations and safety showers.
This guide equips you with the necessary knowledge and practical advice to handle strong acids and bases safely and effectively, whether for academic purposes or professional use. By understanding the underlying principles and following the steps outlined, you can navigate the balancing act of chemistry with confidence and expertise.