Sodium hydroxide, commonly known as caustic soda, plays a critical role in various industrial and household applications. This versatile chemical compound, recognized by its formula NaOH, is indispensable in processes ranging from soap making to heavy-duty cleaning solutions. This article delves into the intricacies of the sodium hydroxide formula, demystifying its structure, uses, and implications in practical applications.
Understanding the Chemical Structure of Sodium Hydroxide
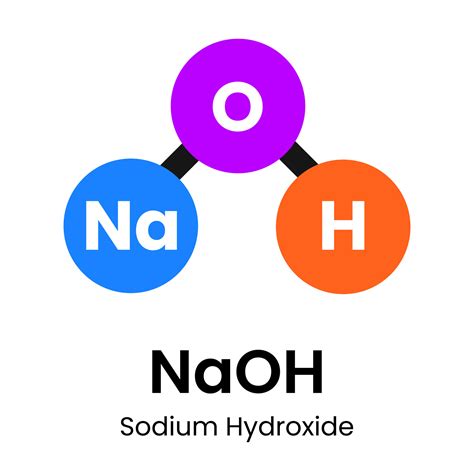
The sodium hydroxide formula, NaOH, represents a quintessential alkali, consisting of a sodium ion (Na+) and a hydroxide ion (OH-). The ionic nature of this compound explains its extensive solubility in water, forming a strongly alkaline solution that can efficiently dissolve oils and fats, thus making it an invaluable component in cleaning products. Its molecular weight is approximately 40 g/mol, a figure critical for calculating appropriate concentrations in various applications.
The Role of Sodium Hydroxide in Industry
In industrial settings, sodium hydroxide is a fundamental chemical in numerous processes. One of its primary roles is in the manufacture of pulp and paper. Here, it is used in the Kraft process for breaking down wood fibers to produce paper. Moreover, NaOH finds application in the production of dyes, fabrics, and even in the manufacture of plastics such as polyvinyl alcohol (PVA). Its utility in these industries underscores the importance of precise control and handling to ensure safety and efficiency.
Key Insights
- Primary insight with practical relevance: Sodium hydroxide's robust alkalinity makes it effective in breaking down complex materials for industrial processes.
- Technical consideration with clear application: Its use in the Kraft process highlights the need for precise handling to ensure both process efficiency and worker safety.
- Actionable recommendation: Always adhere to safety protocols when working with NaOH to prevent accidents and ensure optimal outcomes.
Sodium Hydroxide’s Household and Laboratory Applications
In addition to industrial uses, sodium hydroxide has significant applications in everyday life and academic laboratories. In households, it is a staple ingredient in drain cleaners, acting to dissolve grease and organic matter. The potent alkalinity of NaOH ensures it clears clogs by saponifying fats, making it an essential tool for DIY maintenance. In laboratories, it is indispensable in titration processes to determine the acidity of solutions, facilitating precise chemical analysis.
Safety Considerations When Handling Sodium Hydroxide
Despite its beneficial applications, sodium hydroxide can pose serious health hazards if not handled properly. It is highly corrosive and can cause severe burns upon contact with skin or eyes. When working with NaOH, it is essential to wear appropriate personal protective equipment (PPE) such as gloves, safety goggles, and lab coats. In cases of accidental contact, immediate rinsing with copious amounts of water is crucial to mitigate the effects of the chemical.
Is sodium hydroxide safe to use at home?
While sodium hydroxide can be used safely in household cleaners with proper precautions, it should always be handled with care. Ensure that you are following the instructions on the product label and use protective gear to avoid skin and eye contact.
What should I do in case of NaOH exposure?
If you experience exposure to sodium hydroxide, immediately rinse the affected area with plenty of water for at least 15 minutes. Seek medical attention promptly to assess any potential damage and receive appropriate treatment.
Sodium hydroxide’s formula, NaOH, encapsulates a compound of immense utility across various fields. Its role in industrial manufacturing, its presence in household cleaners, and its importance in laboratory settings underscore its multifaceted nature. However, with great utility comes great responsibility, emphasizing the need for stringent safety protocols when handling this potent chemical. Understanding the chemistry behind NaOH is not just about recognizing its formula but appreciating its broad spectrum of applications and ensuring its safe and effective use in diverse scenarios.