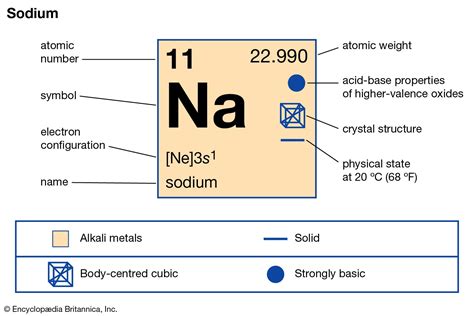
Salt, commonly associated with everyday cooking and table condiments, is deeply rooted in both scientific and culinary traditions. On the periodic table, salt predominantly refers to sodium chloride, an ionic compound of sodium (Na) and chlorine (Cl). It’s essential to understand the chemical nuances and broader implications of salt, as its properties impact not just culinary practices but also industrial processes and health considerations.
Key Insights
- Primary insight with practical relevance: The chemical stability and solubility of sodium chloride provide unique properties that facilitate its use in multiple applications.
- Technical consideration with clear application: Understanding the electrolysis of salt water can lead to the production of essential industrial chemicals like sodium hydroxide.
- Actionable recommendation: For health-conscious individuals, reducing salt intake can significantly lower the risk of hypertension and other cardiovascular issues.
Chemical Properties of Salt
Sodium chloride is an exemplary compound of ionic bonding where sodium, losing an electron, forms a positively charged ion (Na+), while chlorine, gaining an electron, forms a negatively charged ion (Cl-). This ionic interaction results in a highly stable crystal lattice, which accounts for its physical properties like high melting and boiling points. The solubility of sodium chloride in water is due to the interaction between the ions and water molecules, leading to its use in various fields such as medicine, food preservation, and as an antifreeze in winter road treatments.Industrial Applications of Salt
The production of salt is one of the oldest and most widespread industrial processes. One significant industrial application is the electrolysis of brine, which separates sodium chloride into chlorine gas, hydrogen gas, and sodium hydroxide—an essential chemical in manufacturing. This process underscores the versatility of salt beyond culinary use, contributing to sectors like plastics, paper, and cleaning products. Furthermore, sodium chloride is crucial in the chemical industry for processes like water softening and de-icing.Health Implications of Salt Consumption
While salt has indispensable roles across various industries, its consumption in dietary contexts warrants caution. The World Health Organization recommends limiting salt intake to less than 5 grams per day to mitigate risks of hypertension, heart disease, and stroke. High sodium diets can cause the body to retain water, thereby increasing blood volume and putting extra strain on the heart. Therefore, understanding and managing salt intake is crucial for maintaining cardiovascular health.Can all salts on the periodic table be used in cooking?
No, not all salts are safe for culinary use. While sodium chloride is benign and widely used, other salts like sodium fluoride or lead sulfate are toxic and should never be ingested.
What happens if I consume too much salt?
Excessive salt consumption can lead to health problems such as hypertension, heart disease, and stroke, primarily due to the body retaining excess water and increasing blood pressure.
Salt on the periodic table, while commonly perceived as a simple kitchen staple, encompasses a wealth of chemical and practical applications. From its robust role in ionic compound structures to its vital industrial and health implications, sodium chloride epitomizes a compound with far-reaching importance. Balancing its benefits and risks is key to harnessing its potential across various domains effectively.