Welcome to the definitive guide on unlocking the secrets of R and S configuration in chemistry! Many students and professionals find this concept challenging, often leading to confusion and frustration. Our aim is to break down the complexities into clear, actionable advice, ensuring you can understand, apply, and master this fundamental aspect of stereochemistry with confidence and ease. Whether you’re preparing for exams, working on research projects, or just curious about molecular structures, this guide will provide you with the comprehensive knowledge and practical tools you need to tackle R and S configurations head-on.
Understanding the Challenge
R and S configuration is a cornerstone of organic chemistry, yet it’s one of the more perplexing topics many face. The terms R and S refer to the relative spatial arrangements of atoms within chiral molecules, which are molecules that have a non-superimposable mirror image. Understanding these configurations isn’t just about memorizing rules; it’s about grasping the logic behind them. This guide will dissect these configurations into easy-to-follow steps, providing real-world examples and practical solutions to common pitfalls.
Quick Reference
Quick Reference
- Immediate action item: Always assign priority to substituents using the Cahn-Ingold-Prelog (CIP) rules.
- Essential tip: Practice assigning configurations to several chiral molecules to get comfortable with the process.
- Common mistake to avoid: Confusing the terms D/L and R/S; they apply to different contexts and should not be used interchangeably.
Step-by-Step Guidance on Assigning R and S Configuration
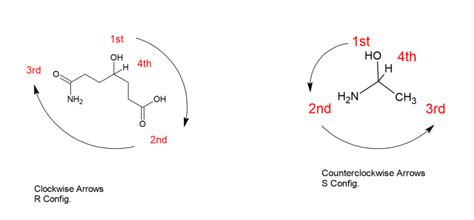
To assign R or S configurations to chiral centers, you need to follow the Cahn-Ingold-Prelog priority rules. Here’s a step-by-step breakdown:
- Identify the chiral center: This is usually a carbon atom with four different groups attached. It must be bonded to four different substituents.
- Assign priorities: According to the CIP rules, assign numbers (1, 2, 3) to the four substituents attached to the chiral center. The priority is based on atomic number; higher atomic numbers get higher priority.
- Orient the molecule: Arrange the molecule in space so that the lowest-priority group (usually hydrogen) is pointing away from you.
- Determine the configuration: Follow the order from highest to lowest priority. If the sequence goes clockwise, the configuration is R (rectus). If it goes counterclockwise, it’s S (sinister).
Here’s a practical example to illustrate these steps:
- Example molecule: Consider 2-bromopentane (C5H11Br). Step 1: Identify the chiral center. Here, it’s the second carbon atom. Step 2: Assign priorities. The priorities are: CH3CH2 (3), CH3 (2), Br (1), CH2CH2CH3 (4). Step 3: Orient the molecule so that the hydrogen is pointing away from you. Step 4: Determine the configuration. The order from highest to lowest priority is: CH3CH2 (1), CH3 (2), Br (3). The sequence goes clockwise, thus the configuration is R.
Practical Application of R and S Configuration
Applying R and S configuration principles to real-world chemistry problems can deepen your understanding and make the concepts more tangible. Let’s explore a couple of practical applications.
In Drug Design
Enantiomers, molecules with the same molecular formula and sequence of bonded atoms but a different three-dimensional arrangement, often exhibit drastically different biological activities. R and S configurations play a critical role in drug design. For example, the drug thalidomide has two enantiomers. One enantiomer was intended to treat morning sickness in pregnant women, but the other caused severe birth defects. Understanding the R and S configurations helps scientists design drugs that target only the desired enantiomer.
In Chiral Synthesis
In chiral synthesis, the goal is to produce one enantiomer in a reaction preferentially over the other. Knowledge of R and S configurations is crucial for planning and executing these syntheses. Consider a synthesis of (S)-ibuprofen, where selectivity for the S-enantiomer leads to the desired biological activity. The chemists’ understanding of stereochemistry allows them to design a reaction pathway that favors the formation of the S-enantiomer.
Practical FAQ
What if two groups have the same priority?
When two substituents have the same priority, you move to the next atoms along the chain until a difference is found. For example, if two groups attached to the chiral center both have CH3 groups, you’ll look at the next carbon to break the tie. If this process does not resolve the priority, you continue this process until a difference is identified. Once you identify the highest priority, you follow the same steps to determine the R or S configuration.
How can I practice assigning configurations?
Practice is key! Start with simple molecules and gradually move to more complex ones. Use practice sets available in textbooks or online. You can also work with molecular models to physically manipulate the molecules and see the spatial arrangement. Flashcards summarizing the CIP rules and common configurations can also be a helpful study tool.
Why is it important to distinguish between R and S configurations?
R and S configurations are critical for understanding the properties of chiral molecules, especially in biological systems where enantiomers often have different effects. For instance, one enantiomer of a drug may be therapeutic while the other could be ineffective or even harmful. Accurate identification ensures the right enantiomer is synthesized and used in medical treatments, impacting patient safety and the efficacy of pharmaceuticals.
This guide serves as a foundational toolkit for navigating the complexities of R and S configurations. By following these steps, you’ll gain not just theoretical knowledge but practical, real-world application skills that are indispensable in the field of chemistry. Whether you’re a student, a researcher, or a professional, mastering these configurations will enhance your problem-solving toolkit and open new avenues for scientific inquiry and innovation. Embrace the challenge, and you’ll find that these secrets of stereochemistry are not only accessible but also incredibly rewarding to unlock.