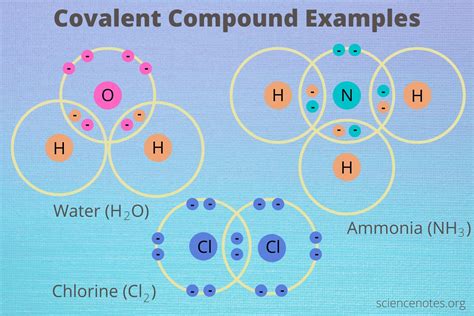
The study of covalent compounds unveils fascinating insights into molecular interactions and bonding, which are essential for understanding chemistry and materials science. Covalent compounds, characterized by the sharing of electron pairs between atoms, have unique properties that influence their behavior, stability, and reactivity. These properties are critical in various applications, from pharmaceuticals to materials engineering.
Key Insights
- Covalent compounds typically exhibit low melting and boiling points.
- The polarity of covalent bonds significantly influences compound solubility and reactivity.
- Designing new covalent compounds can be optimized through computational chemistry.
In understanding covalent compounds, one must recognize the significance of bond strength and the resulting molecular geometry. Bond strength refers to the energy required to break the covalent bond, which varies depending on the atoms involved. For instance, carbon-carbon bonds are generally stronger than carbon-hydrogen bonds, affecting the compound’s stability. Additionally, molecular geometry, determined by the arrangement of atoms around a central atom, impacts properties such as polarity and reactivity. For example, methane (CH4) has a tetrahedral geometry, leading to nonpolar characteristics, while water (H2O) forms a bent geometry, contributing to its polar nature.
Influence of Electronegativity
Electronegativity plays a pivotal role in determining the nature of covalent bonds. This property refers to an atom’s ability to attract electrons in a covalent bond. The difference in electronegativity between two bonded atoms dictates whether the bond is polar or nonpolar. A significant difference results in a polar covalent bond, where the shared electrons are more likely to be found closer to the more electronegative atom. This creates a dipole moment, which is crucial for understanding the compound’s interactions with other molecules. For instance, the difference in electronegativity between hydrogen and oxygen in water results in a polar covalent bond, making water a highly effective solvent.
Solubility and Intermolecular Forces
Covalent compounds also exhibit diverse solubility characteristics based on their polarity and the types of intermolecular forces present. Polar covalent compounds tend to dissolve well in polar solvents like water due to the principle of “like dissolves like.” The presence of dipole moments in these compounds allows them to form hydrogen bonds or dipole-dipole interactions with polar solvents, enhancing solubility. Conversely, nonpolar covalent compounds dissolve well in nonpolar solvents due to similar intermolecular forces. For example, hexane (C6H14), a nonpolar covalent compound, is soluble in nonpolar solvents like benzene but not in polar solvents like water.
Can all covalent compounds form hydrogen bonds?
Not all covalent compounds can form hydrogen bonds. Hydrogen bonds occur when hydrogen is bonded to a highly electronegative atom such as nitrogen, oxygen, or fluorine. Compounds like methane (CH4) cannot form hydrogen bonds as it lacks such electronegative atoms.
How do covalent compounds differ from ionic compounds?
Covalent and ionic compounds differ in their bonding mechanisms. Covalent compounds involve the sharing of electron pairs between atoms, whereas ionic compounds form through the transfer of electrons from one atom to another, resulting in the creation of ions. This fundamental difference influences their physical properties and chemical behaviors.
The exploration of covalent compounds highlights their critical role in both natural and synthetic processes. By understanding their properties, scientists can predict their behavior in various environments, leading to advancements in fields ranging from pharmaceuticals to materials engineering. The strategic design of covalent compounds using computational methods promises to unlock new potentials in technology and medicine.