The potassium sulphate formula is more than just an assortment of symbols. It serves as a pivotal compound in agriculture, industrial processes, and even in environmental science. Delving into the complexities of K2SO4 unveils a wealth of knowledge about its multifaceted applications and benefits. This article aims to demystify the potassium sulphate formula, illuminating its importance through practical insights and evidence-based statements.
Understanding the Potassium Sulphate Formula
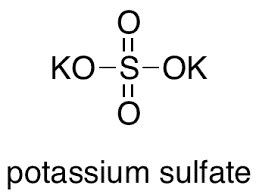
The chemical formula K2SO4 represents potassium sulphate, a salt renowned for its solubility in water and ability to maintain a neutral pH. This compound finds extensive usage in fertilizer formulations due to its ability to supply essential potassium ions to plants, enhancing growth and crop yields. Potassium is crucial in several plant functions, including water regulation, enzyme activation, and overall structural strength. The formula reflects a combination of two potassium (K) atoms, one sulfur (S) atom, and four oxygen (O) atoms, maintaining a balanced ionic structure.
Applications in Agriculture
Potassium sulphate’s role in agriculture is invaluable. It is frequently added to soil as a supplemental fertilizer to rectify potassium deficiencies. This mineral nutrient is particularly vital for fruit and vegetable crops, improving their nutritional value and resistance to diseases. Real-world examples include its incorporation in the cultivation of tomatoes, potatoes, and other high-yield crops. The efficacy of potassium sulphate as a fertilizer is evidenced by studies showing marked improvements in crop quality and yield after its application. Additionally, its use in organic farming aligns with sustainable practices by providing a natural means to enrich soil health without introducing synthetic chemicals.
Key Insights
- Primary insight with practical relevance: Potassium sulphate is essential for enhancing crop yield and quality in agriculture.
- Technical consideration with clear application: Its chemical properties allow it to neutralize soil pH, creating optimal growth conditions.
- Actionable recommendation: Incorporate potassium sulphate in your next fertilization plan for significant agricultural benefits.
Industrial and Environmental Uses
Beyond agriculture, potassium sulphate plays a critical role in various industrial processes. It serves as a key ingredient in the manufacturing of explosives, glass, and other specialty chemicals. In water treatment facilities, it is used to manage water quality and to control the ionic balance, which is vital for maintaining the integrity of filtration systems. Moreover, potassium sulphate contributes to the production of potassium glass, enhancing its transparency and utility in manufacturing high-quality lenses. Environmentally, its non-toxic nature makes it an appealing option for sustainable practices across diverse sectors.
FAQ Section
Is potassium sulphate harmful to plants?
Potassium sulphate is not harmful to plants when used appropriately. It provides essential nutrients without posing a toxicity risk, aiding in balanced growth.
Can potassium sulphate be used in all types of soil?
Yes, potassium sulphate is suitable for various soil types. It neutralizes the pH and supplies necessary potassium, making it versatile for different agricultural settings.
In conclusion, the potassium sulphate formula, K2SO4, is a cornerstone in agricultural and industrial applications. Its multifaceted benefits, from enhancing crop yields to supporting industrial processes, underline its importance. Embracing potassium sulphate in various applications not only boosts efficiency but also aligns with sustainable practices across multiple sectors. Through informed use, we can harness the full potential of this remarkable compound.