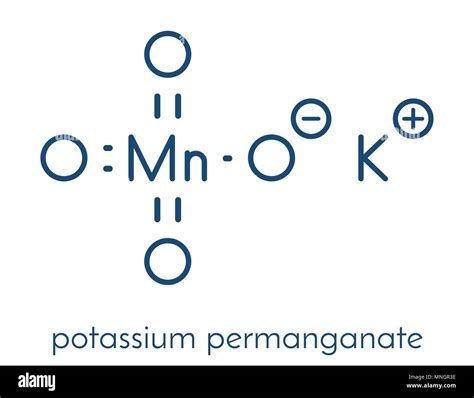
Understanding the potassium permanganate formula is crucial for anyone involved in chemical sciences. This compound, KMnO₄, is widely used in various applications, including redox reactions in laboratories, environmental remediation, and medical treatments. The significance of accurately comprehending the formula cannot be overstated, given its multifaceted roles across different fields.
To grasp the full utility of potassium permanganate, one must dive into the specifics of its chemical composition and properties. This article will provide expert insights, backed by evidence and real-world examples, to demystify the potassium permanganate formula, ensuring professionals and learners alike can utilize it effectively.
The Basic Chemical Makeup
Potassium permanganate is a salt that comprises one potassium (K) atom, one manganese (Mn) atom, and four oxygen (O) atoms, hence its chemical formula KMnO₄. The manganese in potassium permanganate exists in the +7 oxidation state, a highly oxidizing form that makes the compound versatile in its applications.Real-World Application
In chemical reactions, KMnO₄ is frequently used as an oxidizing agent. It oxidizes various substrates, ranging from organic compounds to metals, due to its strong oxidizing nature. For instance, in organic chemistry, potassium permanganate is pivotal in the oxidation of alcohols to carboxylic acids. Its ability to facilitate such transformations makes it an indispensable reagent in synthetic pathways.Critical Considerations for Handling and Usage
When working with potassium permanganate, safety and precision are paramount. The compound is highly reactive, especially in the presence of reducing agents or combustible materials. One must exercise caution when preparing and storing KMnO₄ solutions, as improper handling can lead to hazardous situations. It’s essential to store the compound in a cool, dry place and away from materials that could reduce it.Example in Environmental Science
In environmental science, potassium permanganate has applications in treating contaminated water bodies. Its strong oxidizing properties enable it to neutralize organic pollutants, thereby purifying water supplies. For example, in a polluted river, introducing KMnO₄ can degrade harmful organic compounds, transforming them into less harmful substances through oxidation.Key Insights
- Primary insight with practical relevance: Understanding KMnO₄’s formula is critical for chemists to effectively utilize its strong oxidizing properties in diverse applications.
- Technical consideration with clear application: Always handle KMnO₄ with care due to its strong oxidizing nature to prevent hazardous reactions.
- Actionable recommendation: Ensure proper storage and use of KMnO₄ solutions to maintain its efficacy and safety in various applications.
What happens if potassium permanganate is ingested?
Ingesting KMnO₄ can be highly toxic and may cause severe internal damage. It is a strong oxidizer that can lead to burns and other internal injuries if not handled with extreme caution.
How does potassium permanganate act as an oxidizing agent?
Potassium permanganate acts as a powerful oxidizing agent by accepting electrons from other compounds, thereby causing oxidation reactions. This property makes it highly effective in various chemical synthesis and purification processes.
By delving into the specifics of the potassium permanganate formula, we can appreciate its critical role in numerous scientific fields. With thorough understanding and careful handling, this versatile compound continues to serve as a cornerstone in both laboratory and environmental applications.