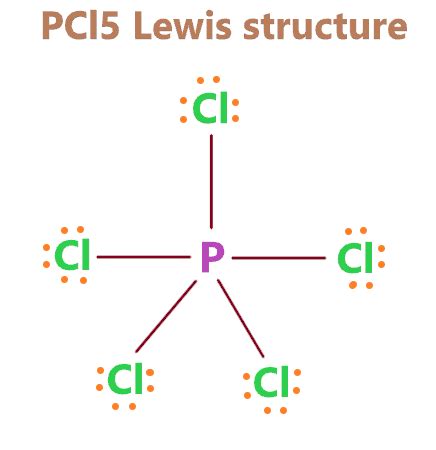
Understanding the PCl5 Lewis structure is pivotal for mastering molecular chemistry, as it offers a foundational comprehension of how atoms bond within a molecule. This insight is not only vital for academic success but also for real-world applications in fields like pharmacology and materials science. The PCl5 molecule, or phosphorus pentachloride, exemplifies the complexity and nuances involved in covalent bonding and molecular geometry.
Key Insights
- PCl5 has an octahedral geometry, contrary to a simple pentagonal planar shape.
- The molecule’s stability arises from the effective use of d-orbitals in bonding.
- Understanding this structure aids in predicting chemical reactivity and molecular behavior.
The Importance of the PCl5 Lewis Structure
The PCl5 Lewis structure is central to understanding its bonding characteristics. Despite its common depiction as a pentagonal planar molecule, PCl5 actually exhibits an octahedral geometry. This revelation is key for chemists when interpreting its chemical properties. The central phosphorus atom forms five covalent bonds with chlorine atoms, but this structure includes an additional complex interaction where phosphorus utilizes its d-orbitals for bonding. This is rare among main-group elements and highlights the broader applicability of molecular orbital theory in organic and inorganic chemistry.Insights into Molecular Geometry and Bonding
In examining the PCl5 Lewis structure, it’s crucial to appreciate the role of phosphorus’s d-orbitals in bonding. Typically, the octet rule confines atoms to form bonds using only s and p orbitals, but phosphorus in PCl5 uses its d orbitals to accommodate five chlorine atoms. This extension challenges traditional bonding theories and illustrates a more complex, hybridized approach. The stability afforded by this configuration means PCl5 is less reactive than its counterparts and can exist as a stable compound at room temperature. This aspect is particularly significant in predicting and manipulating chemical reactions within various industrial and laboratory processes.Why is PCl5's geometry not planar?
PCl5's geometry is not planar due to the use of phosphorus’s d-orbitals in bonding. This allows for an octahedral arrangement, which provides a more stable electron configuration than the typical pentagonal planar shape.
How does the PCl5 Lewis structure influence its chemical reactivity?
The PCl5 Lewis structure, with its stable octahedral geometry, makes the molecule less reactive compared to others that do not utilize d-orbitals. This unique bonding stability is critical for understanding its behavior in chemical reactions.
Understanding the PCl5 Lewis structure offers a gateway to broader chemical principles and their applications. Its complexity illustrates the depth of molecular chemistry and encourages a deeper dive into how advanced bonding theories govern molecular stability and reactivity. The insights gained from studying this molecule can significantly impact fields ranging from drug design to material synthesis, showcasing the practical relevance of this seemingly abstract chemical knowledge.