Understanding PCC Chemistry: A Comprehensive Guide
Are you finding yourself in the complex and often overwhelming world of PCC (Pyridinium chlorochromate) chemistry? If you’re here, it’s likely because you’re looking for that extra edge to elevate your chemical experiments to a higher level. This guide is meticulously crafted to demystify advanced PCC techniques, provide actionable insights, and arm you with the practical knowledge needed to master these powerful yet nuanced chemical reactions.
Problem-Solution Opening Addressing User Needs
PCC chemistry can often seem daunting due to its potential complexity and the necessity for precision. Many chemists find themselves stuck at a crossroads, unsure of how to effectively implement PCC in their advanced reactions. This guide aims to serve as your roadmap through the intricacies of PCC chemistry, from fundamental principles to sophisticated applications. Whether you’re a seasoned chemist seeking to refine your techniques or a newcomer needing a clear and straightforward introduction, this guide will provide you with the practical, expert advice you need to unlock the full potential of PCC reactions.
Quick Reference
Quick Reference
- Immediate action item: Perform a thorough solvent compatibility check before initiating any PCC reaction.
- Essential tip: Always maintain a precise temperature control during the reaction to optimize conversion rates and avoid side reactions.
- Common mistake to avoid: Failing to use an inert atmosphere (e.g., nitrogen or argon) which can lead to oxidation side products.
Mastering Oxidation with PCC
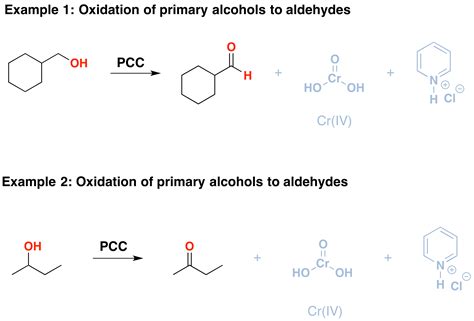
PCC is a highly effective oxidizing agent commonly used for converting primary alcohols to aldehydes and secondary alcohols to ketones. Let’s dive deeper into understanding and mastering this oxidation technique through a detailed, step-by-step process.
Setting Up the Reaction Environment
Before starting your PCC reaction, it’s crucial to prepare the reaction environment correctly. Here are the essential steps:
- Ensure all glassware is dry and clean to avoid dilution or contamination.
- Set up a reaction flask with a magnetic stirrer for efficient mixing.
- Use a round-bottom flask and attach a thermometer to monitor temperature closely.
Preparing the PCC Reagent
PCC is typically prepared in situ to ensure the highest efficiency and to minimize impurities. Here’s a detailed guide:
- Dissolve an appropriate amount of pyridinium chlorochromate in dry dichloromethane (DCM) or another suitable solvent.
- Dissolve the appropriate amount of potassium carbonate (K2CO3) in an aqueous solution.
- Combine the two solutions slowly while stirring to form the active PCC reagent. The ratio is generally PCC to alcohol in a 1:1 molar ratio.
Conducting the Reaction
Now that your environment is set and your reagent prepared, it’s time to conduct the oxidation reaction:
- Add the alcohol substrate slowly to the PCC reagent, maintaining a controlled temperature (usually around 0-5°C).
- Continue stirring the mixture until the reaction is complete, which can usually be monitored by TLC (thin-layer chromatography).
- After the reaction, add a quenching agent such as sodium bisulfite to neutralize excess PCC. Filter the mixture to remove any solid by-products.
Purification and Yield Optimization
To maximize your product yield and purity, follow these steps:
- Extract the product using an appropriate organic solvent like ethyl acetate.
- Wash the organic layer with brine to remove any remaining water-soluble impurities.
- Dry the organic layer over anhydrous sodium sulfate (Na2SO4) and filter.
- Purify the product using column chromatography if necessary.
Advanced PCC Techniques
For those looking to take their PCC chemistry skills to the next level, here are some advanced techniques and insights:
Scaling Up Reactions
When scaling up your PCC reactions from laboratory to industrial scale, attention to detail becomes even more critical:
- Maintain a consistent temperature throughout the reaction using a well-designed jacketed reactor.
- Ensure thorough mixing using efficient impeller designs to avoid local concentration hotspots.
- Utilize automated monitoring systems for real-time control over reaction parameters.
Exploring Alternative Solvent Systems
While dichloromethane is a common solvent for PCC reactions, exploring alternative solvent systems can sometimes yield better results or be more environmentally friendly:
- Investigate the use of polar aprotic solvents like DMSO or DMF, which may enhance reaction rates.
- Evaluate the use of ionic liquids as green alternatives that could potentially improve reaction efficiency.
Optimizing Reaction Conditions
Fine-tuning reaction conditions can greatly affect the efficiency and selectivity of your PCC reactions:
- Experiment with different bases to find the one that gives the best selectivity and yield for your specific substrate.
- Test various temperatures and reaction times to determine the optimal conditions for your reaction.
- Consider using additives like catalytic amounts of transition metals to improve reaction rates or selectivity.
Practical FAQ
What are common pitfalls to avoid when using PCC?
When using PCC, it’s important to avoid common mistakes that can compromise the quality of your reaction. Some of these include:
- Not using an inert atmosphere can lead to oxidation of unwanted side products.
- Overheating the reaction, which can cause degradation of the product or formation of by-products.
- Not controlling the addition rate of the PCC reagent, which can lead to incomplete reactions or over-oxidation.
By carefully controlling reaction conditions and avoiding these pitfalls, you can ensure a successful and efficient PCC oxidation.
Can PCC be used for large-scale industrial processes?
While PCC is powerful and effective for laboratory-scale reactions, its use on an industrial scale requires careful consideration:
- Industrial applications require large quantities of reagents and careful management of waste products.
- Reaction conditions must be optimized for large-scale mixing and temperature control.
- Economic factors and environmental impact must be considered when choosing large-scale reagents and solvents.
However, with the right setup and careful optimization, PCC can certainly be adapted for large-scale industrial processes.
How can one troubleshoot a failed PCC reaction?
Troubleshooting failed PCC reactions involves a systematic approach:
- Check for incomplete reactions by performing a TLC analysis to see if starting materials remain.
- Inspect the temperature control during the reaction to ensure it was maintained within the recommended range.
- Re-evaluate the solvent system and the purity of reagents used, as these can greatly affect reaction efficiency.
- Investigate the possibility of using different bases or additives to improve reaction outcomes.
By following these steps, you can identify and rectify the issues leading to a failed PCC reaction.
Conclusion
PCC chemistry is an indispensable tool in a chemist’s arsenal, offering precise and powerful oxidation capabilities. By understanding and mastering its techniques, you can elevate the quality and efficiency of your chemical processes. This guide has provided you with comprehensive, step-by-step instructions and practical insights to not only handle but excel in PCC chemistry. Armed with this knowledge, you’re well-prepared to unlock the full potential of PCC in your next chemical endeavor.