Understanding ozone (O₃) is crucial for various scientific fields, including atmospheric science and chemistry. Ozone is a molecule made up of three oxygen atoms and plays a significant role in both the Earth’s upper atmosphere and our day-to-day life, from smog formation to ozone layer depletion. This guide will walk you through the intricacies of the ozone Lewis structure, offering practical insights and actionable advice to grasp this concept clearly.
Let's tackle one of the most pressing concerns users often face: struggling to draw the ozone Lewis structure correctly. This guide will provide you with step-by-step guidance to ensure you understand how to represent ozone's molecular structure accurately. Missteps in drawing the Lewis structure can lead to misunderstandings about the molecule's behavior and properties, so getting it right is essential.
Problem-Solution Opening Addressing User Needs
Drawing the ozone (O₃) Lewis structure can be daunting, especially for beginners. The challenge lies in correctly distributing electrons and understanding the molecule's resonance. This guide is designed to demystify the process, offering a straightforward approach to crafting the ozone Lewis structure. By following this guide, you’ll be equipped with the knowledge to visualize ozone’s molecular structure confidently and understand its properties, whether you're a student, a teacher, or a curious chemist.
Why Understanding the Ozone Lewis Structure Matters
Accurately representing the ozone Lewis structure is crucial for several reasons. Firstly, understanding how electrons are shared in a molecule helps in grasping its chemical behavior. Secondly, ozone's structure is pivotal in comprehending atmospheric chemistry and its role in ozone depletion. Lastly, mastering this concept strengthens your foundation in general chemistry, aiding in the understanding of more complex molecules.
Quick Reference
Quick Reference
- Immediate action item: Start with determining the total valence electrons for O₃.
- Essential tip: Use the octet rule to guide your electron distribution, but remember that ozone's structure exhibits resonance.
- Common mistake to avoid: Forgetting that oxygen atoms can share electrons in more than one way, leading to the need for resonance structures.
Detailed How-To Sections
Understanding Ozone’s Valence Electrons
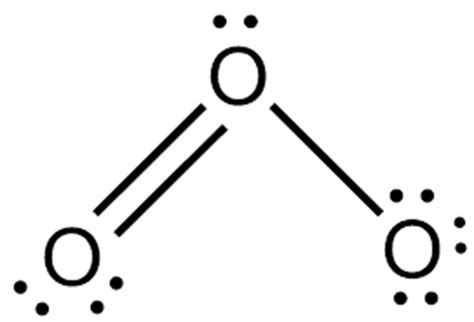
To begin with the ozone Lewis structure, we need to determine the total number of valence electrons. Each oxygen atom has 6 valence electrons, and since there are three oxygen atoms, you multiply 6 by 3 to get 18 valence electrons. This number will guide the distribution process throughout the molecule.
Step-by-Step Distribution
Here’s how you distribute these valence electrons to form the ozone Lewis structure:
- Central Atom Placement: Oxygen, being the same element in all positions in O₃, can be considered as the central atom. Place three oxygen atoms in a triangular arrangement.
- Initial Electron Pair Assignment: Start by giving each oxygen atom two electrons to complete its octet partially. This accounts for 6 electrons, leaving us with 12 electrons to distribute.
- Forming Bonds: Next, create single bonds between the central oxygen atom and each outer oxygen atom. This uses up 6 electrons, leaving us with 6 electrons.
- Completing Octets: The remaining electrons are then distributed around the outer oxygen atoms to complete their octets. Each of the two outer oxygens will now have 8 electrons (4 from bonding and 4 non-bonding).
- Adjusting the Central Atom: The central oxygen atom will end up with 10 electrons in its valence shell because it forms single bonds with each outer atom and still has 2 lone pairs. This is where resonance comes into play.
Resonance in Ozone
Ozone’s structure isn’t as straightforward as initially portrayed due to resonance. The double electron pairs on each outer oxygen atom can shift between them, leading to resonance structures. Here’s how to draw these:
- First Resonance Structure: Place a double bond between the central oxygen and one of the outer oxygens, and single bonds with the other.
- Second Resonance Structure: Shift the double bond to the other outer oxygen, now leaving the first one with a single bond and a lone pair.
- Third Resonance Structure: This mirrors the first structure but with the roles reversed, ensuring both outer oxygen atoms have a double bond at some point.
These resonance structures help explain the molecule's stability and reactivity. Despite the central oxygen atom having 10 electrons, ozone's stability is due to these resonating forms, which average out to give a structure that more closely resembles the real-world molecule.
Common Mistakes and Solutions
When tackling the ozone Lewis structure, there are several common pitfalls. Here are a few to watch out for:
- Incorrect Electron Count: Ensure you correctly count valence electrons. A mistake here will derail your entire structure.
- Overlooking Resonance: Remember, ozone’s stability comes from its ability to resonate. Omitting this step will lead to an incomplete picture.
- Improper Bond Placement: Pay attention to how you distribute bonds and lone pairs, especially around the central atom.
Practical FAQ
Why does the central oxygen in ozone have 10 electrons?
The central oxygen in ozone ends up with 10 electrons due to the nature of resonance in the molecule. Each oxygen atom can share its electrons in multiple configurations to stabilize the molecule. The central oxygen forms two single bonds with the outer oxygens and possesses two lone pairs, totaling 10 electrons in its valence shell.
How does resonance affect ozone’s properties?
Resonance stabilizes ozone, allowing it to have a lower reactivity than would be expected based on its simple Lewis structure. The electrons can move between the oxygen atoms, leading to a delocalization that spreads the molecule’s energy more evenly, contributing to ozone’s chemical behavior.
Can ozone exist without resonance?
From a purely theoretical standpoint, ozone can exist in a single Lewis structure without resonance. However, this structure does not accurately reflect the molecule’s true properties. The real ozone molecule benefits greatly from resonance, which significantly influences its stability and reactivity, making the concept of resonance essential for understanding its behavior.
By understanding and applying the concepts covered in this guide, you can accurately draw the ozone Lewis structure and comprehend its significance in various chemical and environmental contexts.