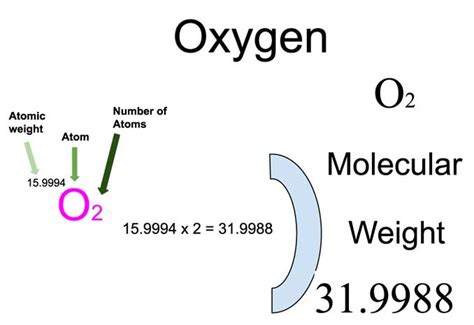Understanding the molar mass of oxygen is vital for various scientific applications, particularly in fields such as chemistry, biochemistry, and environmental science. This piece delves into the essentials of oxygen’s molar mass, offering insights and examples to enhance comprehension.
Key Insights
- Oxygen's molar mass is crucial for stoichiometric calculations in chemical reactions.
- The technical consideration of using oxygen's molar mass affects various fields, from pharmaceutical synthesis to atmospheric science.
- An actionable recommendation is to always double-check the molar mass values when performing any chemical calculation to ensure accuracy.
The Significance of Oxygen Molar Mass
Oxygen, with the molecular formula O₂, is an elemental diatomic molecule whose molar mass is approximately 32 grams per mole. This value is derived from the sum of the atomic masses of two oxygen atoms, each with an atomic mass of about 16 amu (atomic mass units). Accurate determination of molar mass is essential in stoichiometric calculations, which help predict the amounts of reactants and products in chemical reactions. For instance, understanding oxygen’s molar mass allows chemists to accurately calculate the amount of oxygen required in combustion reactions or in the synthesis of various compounds.Applications of Oxygen Molar Mass
In biochemistry, the precise calculation of oxygen’s molar mass is fundamental in metabolic studies. For example, the complete oxidation of glucose (C₆H₁₂O₆) in cellular respiration generates water and carbon dioxide, with oxygen playing a critical role. The balanced chemical equation for this process is:C₆H₁₂O₆ + 6O₂ → 6CO₂ + 6H₂O
To determine the molar amounts of oxygen gas required, the molar mass of oxygen (32 g/mol) is used to convert from mass to moles and vice versa. This precise calculation ensures the correct proportions of reactants, facilitating accurate predictions of energy production in cells.
Advanced Applications in Environmental Science
In environmental science, understanding the molar mass of oxygen is vital for analyzing atmospheric composition and changes. The molar mass is a critical factor in calculating the concentration of oxygen in air samples and determining the impact of pollutants. For example, monitoring the oxygen levels in the atmosphere is essential in assessing air quality and understanding climate change dynamics. By knowing the molar mass of oxygen (32 g/mol), scientists can accurately measure the changes in oxygen concentration due to natural processes or anthropogenic activities, providing valuable data for environmental policy and mitigation strategies.Why is the molar mass of oxygen important in chemical reactions?
The molar mass of oxygen is crucial for stoichiometric calculations. It helps determine the amounts of reactants and products, ensuring accurate predictions in chemical reactions and maintaining the balance of the reaction.
How does the molar mass of oxygen impact metabolic processes?
In metabolic processes, the molar mass of oxygen is fundamental for calculating the proportions of oxygen used in cellular respiration. Accurate values ensure precise predictions of energy production and the correct functioning of biochemical pathways.
This thorough understanding of oxygen’s molar mass underscores its importance across multiple scientific domains, from chemistry and biochemistry to environmental science. By integrating practical insights and real-world examples, one can appreciate the indispensable role of this fundamental property in scientific and environmental studies.