The world of oxidizers is a fascinating and powerful aspect of chemical science, essential for countless industries ranging from manufacturing to environmental management. By understanding the properties and effective use of oxidizers, professionals can unlock enhanced performance and safer processes. This article delves into the practical insights, evidence-based statements, and real-world examples that define the role of oxidizers.
The Fundamentals of Oxidizers
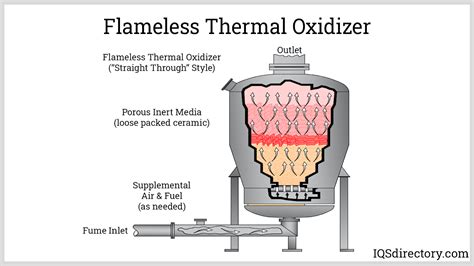
Oxidizers play a critical role in numerous chemical reactions by providing oxygen that facilitates the oxidation process. Typically, these substances are either pure oxygen or compounds containing oxygen that easily release it. This can include substances like hydrogen peroxide, potassium permanganate, and sodium hypochlorite. The potency of an oxidizer directly correlates with its ability to initiate and sustain oxidation, thereby impacting efficiency in processes such as cleaning, bleaching, and combustion.
Key Insights
- Primary insight with practical relevance: Properly managed oxidizers can dramatically enhance industrial cleaning processes, improving both efficiency and safety.
- Technical consideration with clear application: The stability and reactivity of oxidizers must be meticulously controlled to avoid hazardous reactions and ensure safety.
- Actionable recommendation: Regular monitoring and proper handling protocols are essential to mitigate risks associated with oxidizers.
Applications in Industrial Processes
In the industrial sector, oxidizers are indispensable for maintaining high standards of quality and safety. For example, hydrogen peroxide is widely used in the textile industry for its bleaching properties. Its ability to degrade into water and oxygen makes it both effective and environmentally benign. Similarly, potassium permanganate is employed in water treatment for its oxidizing ability to remove organic contaminants.
In manufacturing, oxidizers are crucial for processes like welding and cutting metals, where they enhance flame quality and ensure precision. By integrating oxidizers into these operations, manufacturers can achieve cleaner cuts and more accurate welding, resulting in higher quality products.
Environmental and Safety Considerations
While oxidizers offer numerous advantages, their potential hazards necessitate stringent safety protocols. For instance, sodium hypochlorite, commonly used as a disinfectant in public sanitation, can pose risks if not handled correctly. It can generate chlorine gas in the presence of acids, leading to severe respiratory issues.
To mitigate these risks, it is essential to conduct regular safety assessments and provide adequate training for personnel handling oxidizers. Utilizing appropriate personal protective equipment (PPE) and ensuring proper ventilation in work areas can greatly reduce exposure risks. Additionally, understanding the chemical compatibility of oxidizers with other materials in the environment is crucial for preventing accidental reactions.
What are the primary hazards associated with oxidizers?
The primary hazards of oxidizers include the potential for explosive reactions with reducing agents, toxic gas generation, and respiratory risks if not handled properly.
How can industries safely manage oxidizers?
Industries can safely manage oxidizers by implementing strict handling protocols, providing comprehensive training for employees, using proper PPE, and ensuring adequate ventilation in workplaces.
In summary, oxidizers are powerful tools that, when correctly managed, can greatly enhance industrial processes. Understanding their properties and implementing safety measures ensures both efficiency and protection in various applications.