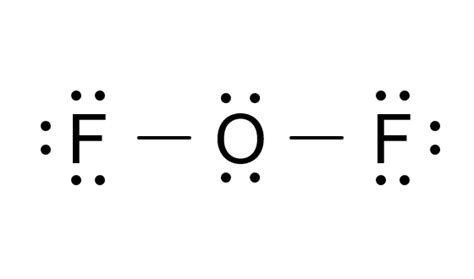Unlocking the secrets of OF2 (oxygen difluoride) Lewis structure can be an intriguing venture for chemists and learners alike. Understanding the correct placement of atoms, lone pairs, and bond formations provides valuable insight into the compound’s molecular behavior and stability. This article aims to elucidate the critical aspects of the OF2 Lewis structure, offering expert insights, real-world examples, and practical recommendations.
Introduction to OF2
Oxygen difluoride (OF2) is a chemical compound consisting of one oxygen atom and two fluorine atoms. Its structure is fundamental to grasping its reactivity, bonding behavior, and overall molecular geometry. As with any molecule, comprehending its Lewis structure is crucial for predicting its chemical properties and interactions.Key Insights
Key Insights
- OF2 has a bent molecular geometry due to the lone pairs on the central oxygen atom.
- The oxygen atom in OF2 forms two single bonds with the fluorine atoms, showcasing sp3 hybridization.
- Correctly depicting lone pairs on the oxygen atom is essential for understanding its polarity and reactivity.
The Lewis Structure Composition
The Lewis structure of OF2 is an excellent example to study valence electrons and molecular bonding. Oxygen has six valence electrons, and each fluorine atom contributes seven. Therefore, the total valence electrons in OF2 are 16. To construct the Lewis structure, oxygen should form two single bonds with the two fluorine atoms. This accounts for four valence electrons. The remaining twelve electrons are distributed as lone pairs—six on the oxygen atom (forming three lone pairs).In this setup, oxygen ends up with a total of eight electrons around it, adhering to the octet rule, while fluorine completes its octet with eight electrons each. The distribution results in a bent shape, indicative of the two lone pairs repelling the single bonds and altering the bond angle from the typical 109.5 degrees in sp3 hybridization.
Electronegativity and Polarity
Electronegativity is a vital factor in determining the polarity of OF2. Oxygen and fluorine have significant differences in electronegativity; fluorine is more electronegative than oxygen. Consequently, the electron pairs are pulled more towards the fluorine atoms, causing a dipole moment. Despite the central oxygen atom having lone pairs, the overall molecule exhibits polarity because the bond dipoles do not cancel each other out perfectly due to the bent shape. This molecular polarity significantly influences its reactivity and interactions in chemical reactions.Hybridization in OF2
Hybridization is a critical concept in molecular chemistry. In OF2, the oxygen atom undergoes sp3 hybridization. This hybridization allows the formation of four sp3 hybrid orbitals, enabling two to form sigma bonds with the fluorine atoms, while the other two accommodate the lone pairs. This hybridized state accounts for the molecule’s bent geometry, affecting its physical and chemical properties, including its reactivity in various reactions.FAQ Section
What is the bond angle in OF2?
The bond angle in OF2 is less than 109.5 degrees due to the repulsion from the lone pairs on the oxygen atom, resulting in a bent molecular geometry.
Is OF2 polar or nonpolar?
OF2 is polar due to its bent geometry and the significant difference in electronegativity between oxygen and fluorine.
Understanding the OF2 Lewis structure and its implications on molecular geometry, hybridization, and polarity provides a comprehensive overview of this fascinating compound. These insights are invaluable for anyone delving into the realms of molecular chemistry and chemical bonding.