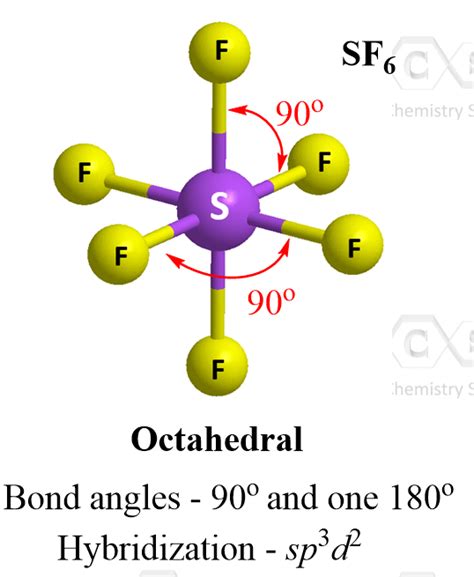
The octahedral geometry, a fundamental shape in coordination chemistry, remains one of the most intriguing and well-studied structures in molecular science. The octahedral bond angle, specifically the 90-degree angle between ligands, is not just an abstract concept but a practical cornerstone that influences molecular stability, reactivity, and electronic properties. This article delves into the core principles and real-world examples that illustrate the significance of octahedral bond angles.
Key Insights
- Primary insight with practical relevance: Understanding the 90-degree bond angles in octahedral complexes aids in predicting molecular behavior and interactions.
- Technical consideration with clear application: The specific spatial arrangement of ligands in octahedral geometry influences the ligand field splitting, which is crucial in understanding spectroscopic and magnetic properties.
- Actionable recommendation: Chemists should employ computational modeling to visualize and analyze the octahedral structures for a deeper understanding of their chemical behavior.
Theoretical Foundation
The concept of octahedral geometry is deeply rooted in the principles of VSEPR (Valence Shell Electron Pair Repulsion) theory. According to VSEPR, electrons surrounding a central atom repel each other, thereby arranging themselves to achieve maximum distance, resulting in the formation of a regular octahedron where the bond angles are precisely 90 degrees. This geometry is predominant in six-coordinate complexes, such as in the case of [Fe(H2O)6]2+, where the iron ion is surrounded by six water molecules. The 90-degree bond angles facilitate an optimal distribution of the electronic cloud, minimizing repulsive forces between the electron pairs.
Real-World Applications
The ramifications of octahedral bond angles extend beyond pure theory and manifest in practical applications across various fields. For instance, in the realm of materials science, transition metal complexes often adopt octahedral geometries, which play a crucial role in the functionality of catalysts. In the Haber-Bosch process, the ammonia synthesis catalyst relies on iron complexes with octahedral configurations to facilitate the conversion of nitrogen and hydrogen gases into ammonia. The 90-degree angles within these complexes determine the ligand’s field splitting and, consequently, the energy levels available for electron transitions, which are essential for the catalyst’s efficiency.
Another tangible example is in the field of medicinal chemistry, where certain metal complexes with octahedral coordination are used in pharmaceuticals. An eminent example is cisplatin, a platinum-based chemotherapy drug, which exhibits an octahedral structure. The spatial arrangement and bond angles enable cisplatin to bind to DNA in cancer cells, thereby inhibiting replication and leading to cell death. The understanding of these octahedral geometries at a molecular level allows chemists to modify and design more effective therapeutic agents.
How do octahedral bond angles affect the chemical reactivity of a complex?
Octahedral bond angles impact the ligand field splitting, which determines the energy gap between the d-orbitals of the central metal ion. This gap influences the complex's reactivity by affecting the ease with which electrons can move between orbitals during chemical reactions. A larger gap can make the complex less reactive, whereas a smaller gap facilitates easier electron transitions and increased reactivity.
Can distortions occur in octahedral geometries?
Yes, deviations from the ideal octahedral geometry can occur due to various factors such as steric strain or electronic effects. These distortions can be classified into two main types: Jahn-Teller distortions, where an elongation or compression of the octahedron leads to lowering of symmetry, and structural distortions where the bond angles are altered due to ligand interactions.
In conclusion, the octahedral bond angle’s precise 90-degree measure is more than a theoretical construct; it’s a critical parameter in understanding and predicting the behavior of complex molecules in diverse chemical applications. Through this knowledge, chemists can innovate, optimize, and synthesize novel compounds for technological advancements and therapeutic breakthroughs.