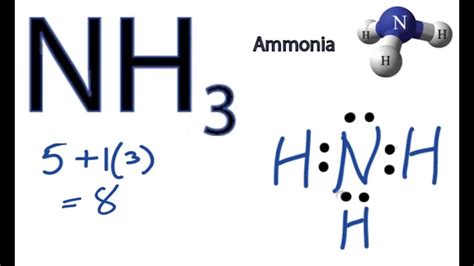NH3 Lewis Dot Structure Breakdown Unveiled!
Understanding the NH3 Lewis dot structure is crucial for mastering concepts in chemistry. The ammonia molecule (NH3) is a fundamental compound encountered in various chemical contexts, including general chemistry, biochemistry, and more advanced fields like organic chemistry. This guide is designed to provide a thorough and practical breakdown of the NH3 Lewis dot structure, guiding you from basic understanding to expert insight. By following this step-by-step guidance, you will gain actionable advice, practical solutions, and expert tips to enhance your comprehension and application of Lewis dot structures.
The NH3 molecule, consisting of one nitrogen atom bonded to three hydrogen atoms, provides an excellent platform for learning about Lewis dot structures. Many students face challenges in grasping how to accurately represent the bonding and lone pairs in NH3. This guide aims to address these pain points and offer a straightforward, problem-solving approach.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Begin with the central atom (Nitrogen) and its valence electrons. Nitrogen has 5 valence electrons.
- Essential tip with step-by-step guidance: Distribute the 8 total valence electrons (5 from Nitrogen + 3 from Hydrogen atoms) around the central atom to satisfy the octet rule.
- Common mistake to avoid with solution: Don’t forget to place lone pairs on the central atom to fulfill its valence shell, making sure it has 8 electrons around it.
This guide will break down the steps to construct the NH3 Lewis dot structure, providing real-world examples and practical solutions. We’ll cover everything from basic drawing techniques to advanced tips that will help solidify your understanding.
Detailed How-To Sections
Step-by-Step Guide to Drawing the NH3 Lewis Dot Structure
Constructing the Lewis dot structure for NH3 requires several steps. Let’s go through each step meticulously:
- Identify the Central Atom: In NH3, the central atom is Nitrogen (N). This element forms the backbone of the molecule, around which the three hydrogen atoms (H) will bond.
- Count Valence Electrons: Determine the total number of valence electrons for the molecule. Nitrogen has 5 valence electrons, and each hydrogen has 1, so we have:
- 5 (from Nitrogen) + 3*1 (from Hydrogen) = 8 valence electrons in total.
- Arrange Atoms: Place the nitrogen atom in the center, with the three hydrogen atoms surrounding it.
- Bond Formation: Connect each hydrogen to the nitrogen atom with single bonds. Each bond uses 2 electrons (one from each atom).
- Each hydrogen forms one bond with the nitrogen.
- This uses up 3 of our 8 valence electrons (2 per bond, 3 bonds = 6 electrons).
- Distribute Remaining Electrons: After forming bonds, there are 2 electrons left, which need to be distributed to the central nitrogen atom to complete its octet.
- Place the remaining electrons in pairs around the nitrogen atom.
- This gives nitrogen 8 electrons, fulfilling the octet rule.
Tips, Best Practices, and Common Mistakes
Here are some tips to help you accurately draw the NH3 Lewis dot structure:
- Tip 1: Always start with the central atom and ensure it’s surrounded by the atoms it bonds with.
- Tip 2: Count the valence electrons at the start to ensure you don’t overshoot or undershoot in your structure.
- Tip 3: Follow the octet rule but remember that the central atom sometimes can have fewer than 8 electrons if it’s bonded to fewer atoms.
- Best Practice: Check your final structure to ensure all atoms have fulfilled their valence shell (except hydrogen, which follows the duet rule with 2 electrons).
- Common Mistake to Avoid: Forgetting to place lone pairs on the central atom or incorrectly distributing electrons.
Understanding the above steps will greatly enhance your ability to construct accurate Lewis dot structures.
Practical FAQ
What if I can't seem to get the NH3 structure right?
If you're struggling with constructing the NH3 Lewis dot structure, it’s often due to misunderstanding the valence electrons and bonding rules. Revisit basic concepts such as how to count valence electrons and how atoms bond. Use diagrams and practice with simpler molecules before tackling NH3. Additionally, ensure you’re placing lone pairs correctly on the central atom to satisfy its valence shell requirements.
Why is it important to ensure NH3 has a complete octet around the central nitrogen atom?
The octet rule is a fundamental principle in chemistry that suggests atoms are most stable when they have eight electrons in their valence shell. By ensuring nitrogen (and other atoms) in NH3 satisfies the octet rule, the molecule becomes more stable and less reactive. This stability is critical in predicting molecular behavior in chemical reactions.
Can NH3 have different structural configurations?
NH3 is structurally simple and does not exhibit isomerism (different structural configurations with the same molecular formula). The ammonia molecule is always trigonal pyramidal in shape due to the single central nitrogen atom bonded to three hydrogen atoms and with one lone pair of electrons on nitrogen.
By following this detailed guide, you’ll gain confidence in drawing and understanding the NH3 Lewis dot structure. Always remember to take it step by step and practice regularly to reinforce your learning. Whether you’re a beginner or an advanced learner, this guide provides the necessary tools and insights to master NH3 Lewis dot structures.
Through this guide, you’ve learned how to approach the NH3 molecule from an understanding of its components to constructing its Lewis dot structure. With practice and the tips provided, you can confidently apply these techniques to other molecules and deepen your knowledge in chemistry.