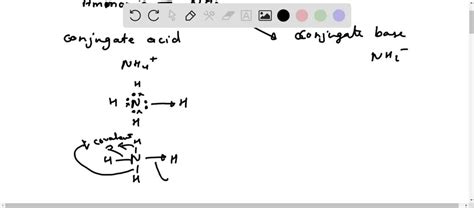
Ammonia, or NH3, is a compound that often stirs curiosity due to its dual nature in the realms of chemistry. To demystify its classification, we explore its behavior as both an acid and a base in various contexts. This discussion aims to provide a comprehensive understanding of NH3’s properties, guided by practical insights and real-world examples.
The Chemistry Behind NH3
Ammonia, NH3, is a colorless gas that is highly soluble in water. When dissolved in water, it undergoes a unique reaction that illustrates its base behavior:
NH3 + H2O → NH4+ + OH-
This reaction reveals ammonia’s ability to accept protons (H+), which is the hallmark of a base according to the Brønsted-Lowry definition. By producing hydroxide ions (OH-) in water, ammonia exemplifies a basic compound.
NH3 as a Bronsted-Lowry Base
A closer look at NH3 through the lens of the Bronsted-Lowry theory highlights its role as a base. Here, a base is defined as a proton acceptor. In its interaction with acids, ammonia demonstrates its ability to neutralize them:
For instance, when NH3 encounters hydrochloric acid (HCl), it forms ammonium chloride (NH4Cl) as follows:
NH3 + HCl → NH4Cl
This example reinforces NH3’s behavior as a base by accepting the H+ ion from the acid.
NH3’s Acidity in Different Contexts
Though primarily recognized as a base, NH3 exhibits acid characteristics under certain circumstances. Specifically, in a medium that doesn’t include water, NH3 can donate a pair of electrons rather than a proton, akin to Lewis acid behavior. Here’s an example with boron trifluoride (BF3):
NH3 + BF3 → F3B-NH3
In this reaction, NH3 acts as a Lewis base by donating a lone pair of electrons to BF3, which functions as a Lewis acid.
Key Insights
- NH3 is predominantly recognized as a base due to its ability to accept protons.
- It can act as a Lewis base by donating a lone pair of electrons in contexts without water.
- The practical application of NH3 as a buffering agent in industrial processes showcases its versatility.
Is NH3 always a base?
No, while NH3 commonly acts as a base due to its proton-accepting behavior, it can also exhibit Lewis base characteristics in appropriate contexts.
What industries use NH3 as a base?
Industries such as agriculture (as a fertilizer) and pharmaceuticals use NH3 in its basic form for various applications.
In summary, NH3’s chemical behavior is multifaceted, serving as a fundamental base while also demonstrating nuances of acidity. Understanding its dual nature allows for a broader application in science and industry. Whether neutralizing acids or participating in electron-pair interactions, ammonia stands as a pivotal compound in diverse chemical processes.