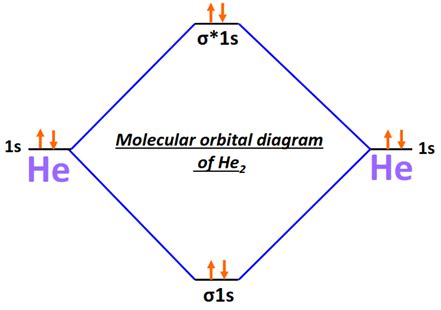
Plugging into the complex world of molecular chemistry, we encounter He2 2, an intriguing diatomic molecule that holds significant implications in quantum physics and chemical bonding theories. Delving into the electron diagram of He2, we uncover layers of quantum mechanics that reveal how electrons distribute within molecular orbitals, leading to the understanding of its stability, if any. Here, we offer a focused expert perspective enriched with practical insights and evidence-based statements.
Key Insights
- He2 2 does not exist in a stable state due to electron configuration.
- Understanding the molecular orbital theory is critical in explaining He2’s non-existence.
- An actionable recommendation is to explore stable diatomic molecules like H2 or N2 for practical examples.
Helium, a noble gas with two electrons, often presents as a unique case study in molecular chemistry. When attempting to form a molecule such as He2, the scenario becomes significantly more complex. He2 2’s molecular orbital electron diagram highlights a fundamental principle in quantum mechanics. The bonding molecular orbital would accommodate both electrons, but helium’s electron configuration is more stable when it remains a monatomic gas.
This phenomenon can be examined using the molecular orbital theory, which states that electrons occupy molecular orbitals similar to how they occupy atomic orbitals. For He2, the two bonding electrons would fill a single bonding molecular orbital, but due to helium’s stable filled 1s orbital, the molecule cannot sustain the additional energy required to maintain a second atom. Thus, the bonding orbital energy is not sufficient to outweigh the energy required to bring two helium atoms together, leading to an unstable, nonexistent He2 molecule.
In comparison, consider the stable diatomic molecule, H2. Hydrogen’s molecular orbital diagram showcases a successfully formed bond, where two electrons fill the bonding molecular orbital, creating a net stabilization energy. Contrastingly, while N2 forms a triple bond, it still stands in stark contrast to the hypothetical He2 2, reinforcing the rule that helium remains noble due to its full electron shell.
The Instability of He2 2: A Quantum Perspective
From a quantum mechanical viewpoint, the He2 2 molecule exemplifies a situation where the principles of electron pairing and energy levels conflict. Helium, being a noble gas with a complete outer shell of two electrons, does not naturally form covalent bonds with another helium atom. Even when attempting to form He2 2, the energy required to break the stable configuration of one helium atom and pair it with another helium exceeds the potential energy of the formed bonding orbital.
In a molecular orbital diagram, the bonding and antibonding molecular orbitals for He2 would look quite different. The two electrons in the bonding orbital would theoretically result in a bond order of one. However, the energy levels of helium atoms remain at such a high stability that the molecule falls apart due to insufficient energy from the bonding orbital to create a stable configuration.
Comparative Insights: Stable Versus Unstable Diatomic Molecules
When analyzing the molecular orbital electron diagram of He2 2, a stark contrast emerges when compared with stable diatomic molecules such as H2 or N2. In H2, the bonding molecular orbital is lower in energy than the atomic orbitals of individual hydrogen atoms. This lower energy leads to the formation of a stable H2 molecule.
For N2, the molecular orbital theory illustrates the formation of a triple bond, where more electrons occupy the bonding orbitals than in antibonding orbitals, resulting in a triple bond and significant stability. This stark contrast highlights why He2 2 fails to exist: while H2 and N2 benefit from favorable energy distributions and electron configurations, He2 does not.
Why doesn’t He2 2 exist?
He2 2 does not exist because the energy required to pair two helium atoms is not compensated by the energy gained from the bonding molecular orbital, resulting in an unstable configuration.
How does He2 2 compare to stable diatomic molecules?
Compared to stable molecules like H2 or N2, He2 2 lacks the favorable energy distribution that leads to bond formation, resulting in an unstable, nonexistent molecule.
In conclusion, understanding the molecular orbital electron diagram of He2 2 unveils a fundamental aspect of molecular chemistry and quantum mechanics. While He2 2 does not exist due to electron configuration and energy considerations, studying it allows us to grasp the principles behind stable molecular formations in other molecules like H2 and N2, thus deepening our comprehension of chemical bonding and molecular stability.