Understanding the molar mass of hydrogen gas (H₂) is pivotal in both academic and professional chemistry fields. The concept forms the foundation for grasping chemical reactions, stoichiometry, and various molecular calculations. Here, we delve into the practicalities, technicalities, and significance of calculating the molar mass of H₂ in a straightforward manner, ensuring comprehension and application.
Key insights
Key Insights
- Molar mass calculation is crucial for predicting chemical reactions’ outcomes.
- Understanding H₂’s molar mass aids in precise dosages in industrial and laboratory settings.
- An actionable recommendation: Always verify molar mass values using the most current atomic weight data.
The foundation of chemistry hinges on the concept of molar mass, representing the mass of one mole of a substance. For diatomic hydrogen (H₂), the calculation begins with the fundamental understanding that one mole of any substance contains Avogadro’s number (6.022 x 10²³) of entities. To find H₂’s molar mass, one must sum the atomic masses of its constituent atoms.
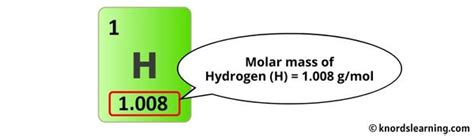
Historically, the atomic mass unit (amu) has been replaced by the unified atomic mass unit (u), where 1 u equals 1⁄12 the mass of a carbon-12 atom. For hydrogen, the atomic mass is approximately 1.008 u. Since H₂ consists of two hydrogen atoms, the molar mass of H₂ is calculated as 2 x 1.008 u, equating to 2.016 u. Converting to grams per mole, this translates to 2.016 g/mol. This simple calculation has profound implications, from determining reaction yields to ensuring precise chemical formulations in industrial processes.
Calculating Molar Mass: A Step-by-Step Approach In chemistry, accurately determining the molar mass of any molecule is essential for understanding its properties and behavior in various reactions. The molar mass serves as a bridge between the macroscopic world we observe and the microscopic realm of atoms and molecules. When calculating the molar mass of H₂, we follow a systematic approach grounded in fundamental principles. Firstly, we identify the number of each type of atom in the molecule. For H₂, there are two hydrogen atoms. Next, we refer to the periodic table to find the atomic mass of each hydrogen atom, which is approximately 1.008 u. The final step involves multiplying the number of atoms by their respective atomic masses and summing the results to get the molar mass of the compound.
This step-by-step process ensures accuracy and consistency in scientific calculations, enabling chemists to predict reaction outcomes, determine necessary quantities of reactants, and understand the behavior of gases under different conditions.
Industrial Applications of Molar Mass Calculations Accurate molar mass calculations are not just academic exercises; they have significant industrial applications. For example, in the production of ammonia (NH₃) via the Haber process, precise calculations of molar masses are crucial for optimizing the ratio of hydrogen to nitrogen gases and ensuring the efficiency of the reaction. Miscalculating the molar mass can lead to incorrect stoichiometric ratios, resulting in reduced yields and increased costs. Thus, understanding and applying the molar mass of H₂ and other gases in industrial processes is fundamental to operational efficiency and economic viability.
FAQ Section
Why is molar mass important in chemical reactions?
Molar mass is essential for determining the stoichiometry of chemical reactions, which in turn dictates the proportion of reactants needed and the quantity of products formed. It enables precise measurements, ensuring the reactions proceed as expected and efficiently.
How do periodic table updates affect molar mass calculations?
Periodic table updates, especially those reflecting new atomic mass values, necessitate recalculating molar masses. Keeping atomic mass data current ensures the accuracy of scientific calculations and predictions in chemical processes.
Grasping the molar mass of H₂ enhances one’s ability to tackle more complex chemical calculations and understand the broader applications of chemistry in various fields. By mastering this fundamental concept, students and professionals alike can unlock deeper insights into chemical reactions and their practical implications.