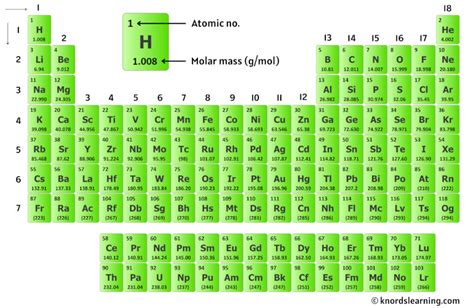
Understanding molar mass is a crucial skill for chemistry enthusiasts. This foundational concept underpins a wide range of applications, from predicting chemical reactions to calculating the necessary reagents for laboratory experiments. This article demystifies molar mass and equips you with the knowledge to master it effortlessly.
Key insights box:
Key Insights
- Molar mass is the mass of a given substance per mole of a chemical compound, expressed in grams per mole (g/mol).
- It is essential for stoichiometric calculations in chemistry to determine the relationship between reactants and products.
- To calculate molar mass, sum the atomic masses of all atoms present in the chemical formula.
Molar mass is a fundamental property that bridges the gap between the atomic world and macroscopic measurements. It represents the mass of one mole of a given substance and is typically expressed in grams per mole (g/mol). This parameter is critical in chemical equations, allowing chemists to convert between mass and the number of moles, facilitating stoichiometric calculations essential for determining reaction yields and quantities of reactants and products.
Molar mass calculation stands as a cornerstone of chemistry, intertwining elements of both chemistry and mathematics. To compute the molar mass of a compound, you need to sum the atomic masses of all atoms in its chemical formula. For instance, the molar mass of water (H₂O) is calculated by adding the atomic masses of two hydrogen atoms and one oxygen atom. Hydrogen’s atomic mass is approximately 1 g/mol, and oxygen’s is around 16 g/mol. Thus, the molar mass of H₂O is 2(1) + 16 = 18 g/mol. This straightforward process enables accurate predictions of chemical reactions’ scales and outcomes.
While the calculation process might seem rudimentary, its implications are profound. For example, when balancing chemical equations, understanding and accurately determining molar mass ensures that the quantities of reactants and products align with the principles of stoichiometry. A slight miscalculation can lead to incorrect predictions, demonstrating the critical nature of precision in chemistry.
Let’s delve deeper into another practical application. Consider the compound sodium chloride (NaCl). Sodium has an atomic mass of approximately 23 g/mol, and chlorine’s is about 35.5 g/mol. Therefore, the molar mass of NaCl is 23 + 35.5 = 58.5 g/mol. This calculation aids in determining how much sodium chloride to use in various reactions, such as creating a buffer solution in biochemistry.
Understanding the concept of molar mass also allows chemists to perform mole-to-mass conversions effortlessly. For example, knowing the molar mass of carbon dioxide (CO₂), which is 44 g/mol, helps a scientist determine how many grams of CO₂ are produced from a given number of moles. This calculation is invaluable in fields like environmental science, where monitoring CO₂ levels is essential.
FAQ section:
What is the difference between molar mass and molecular weight?
Molar mass and molecular weight are essentially the same concept. Both refer to the mass of a mole of a substance. The term’molecular weight’ is often used for individual molecules, whereas’molar mass’ is typically used in the context of chemical formulas and compounds.
Why is molar mass important in chemical reactions?
Molar mass is crucial in chemical reactions because it helps determine the quantitative relationships between reactants and products. It allows chemists to perform accurate stoichiometric calculations, ensuring that the correct proportions of substances are used in reactions, which is vital for both laboratory work and industrial processes.
In conclusion, mastering the concept of molar mass empowers chemistry enthusiasts to navigate the intricacies of chemical reactions with confidence. By grasping how to calculate and apply molar mass, you unlock a deeper understanding of chemical behavior and improve the precision and efficiency of your experiments.