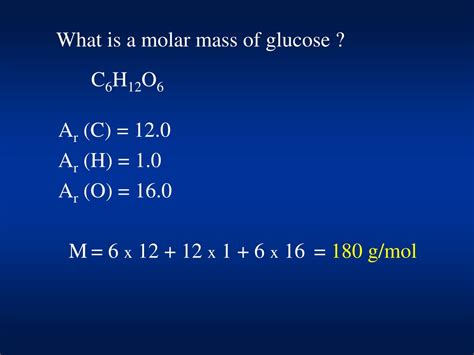Plunging into the heart of biochemistry, it is paramount to understand the fundamental concepts that govern the molecular underpinnings of living organisms. One such critical concept revolves around the molar mass of glucose, a sugar molecule fundamental to cellular metabolism. This deceptively simple compound, C₆H₁₂O₆, carries profound implications for both theoretical and applied sciences. By decoding its molar mass, we uncover insights that resonate far beyond mere academic curiosity.
Key Insights
- Understanding the molar mass of glucose is crucial for applications in biochemistry, nutrition, and pharmaceuticals.
- Accurate calculations of molar mass allow for precise formulations in metabolic pathways and chemical reactions.
- Knowledge of glucose’s molar mass supports practical applications in diagnosing and managing diabetes.
To appreciate the molar mass of glucose, we first need to dissect its molecular structure. Glucose consists of six carbon © atoms, twelve hydrogen (H) atoms, and six oxygen (O) atoms. The molar mass is an aggregate of the atomic masses of each element. Carbon, weighing 12.01 grams per mole, contributes 12.01 * 6 = 72.06 grams. Hydrogen, with an atomic mass of 1.008 grams per mole, contributes 1.008 * 12 = 12.096 grams. Oxygen, with an atomic mass of 16.00 grams per mole, contributes 16.00 * 6 = 96.00 grams. Adding these values together, the molar mass of glucose equals 72.06 + 12.096 + 96.00 = 180.156 grams per mole.
Significance in Biochemistry
In biochemistry, glucose stands as a primary energy source for cellular respiration. The detailed understanding of its molar mass is essential for accurately modeling metabolic pathways. For instance, glucose-6-phosphate is a critical intermediate in glycolysis, the process by which cells convert glucose into energy. The precise molar mass aids in calculating the yield and efficiency of biochemical reactions.Applications in Clinical Nutrition
Beyond laboratories and research settings, glucose’s molar mass has practical implications in clinical nutrition and metabolic health. For example, when calculating the caloric intake of a diet, understanding the precise mass of glucose molecules helps in accurate nutritional planning. This knowledge is particularly significant in managing conditions like diabetes mellitus, where blood glucose levels must be meticulously monitored.FAQ Section
Why is the molar mass of glucose important?
The molar mass of glucose is crucial for accurately calculating the energy and nutrient content in various biochemical and nutritional contexts. It is essential for designing effective metabolic treatments and understanding energy utilization by cells.
How does glucose's molar mass affect its role in the body?
The molar mass of glucose directly influences its role as an energy source. It helps in determining how much energy can be derived from glucose metabolism, which is vital for both physiological functions and clinical applications like metabolic disorder management.
By elucidating the molar mass of glucose, we unlock a deeper understanding of its functional and therapeutic roles. This comprehension not only enriches scientific knowledge but also paves the way for advanced research and practical health applications. It is a reminder of how fundamental chemical principles serve as the bedrock for both life science advancements and medical breakthroughs.