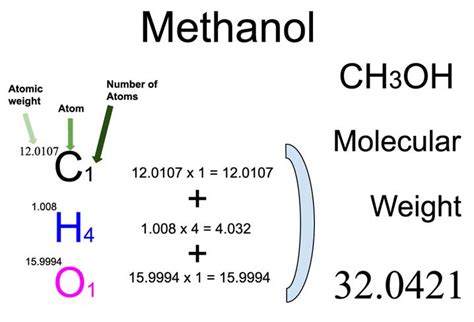
Understanding methanol is pivotal for numerous industries, from fuel to chemical synthesis. With a molecular weight of 32.04 g/mol, methanol’s simplicity belies its vast array of applications. This article delves into its molecular makeup, explores its importance, and provides practical insights into handling this versatile compound.
Key insights box:
Key Insights
- Methanol’s molecular weight of 32.04 g/mol underscores its simplicity yet significance.
- Understanding methanol’s role in fuel cell technology highlights a technical consideration for future energy solutions.
- Adherence to safety protocols when handling methanol is an actionable recommendation for all professionals.
The essence of methanol’s molecular structure revolves around its single carbon atom, four hydrogen atoms, and one oxygen atom. Its molecular formula, CH₃OH, indicates the formation of a methyl group (CH₃) and a hydroxyl group (OH), combining to form methanol. The relatively light molecular weight is indicative of methanol’s fluid properties, which play a crucial role in its application across various industries. From a practical perspective, methanol’s low viscosity and high solubility in water facilitate its use in antifreeze formulations and solvents.
In analyzing methanol’s role in industrial chemistry, one must highlight its significance in producing other compounds, including formaldehyde and acetic acid. The catalytic processes involved in converting natural gas to methanol epitomize its importance in modern chemical production. This transition is primarily due to methanol’s capability to act as a feedstock for synthesizing higher-value chemicals. The molecular simplicity of methanol does not detract from its value in facilitating complex chemical transformations.
Methanol also stands out in the realm of alternative energy solutions. Its potential as a hydrogen carrier in fuel cells merits attention due to its ability to store and release hydrogen upon combustion. This feature provides an essential pathway for clean energy transport and storage. The molecular weight of methanol influences its energy density and efficiency in fuel cell applications, making it a competitive candidate in the ongoing quest for sustainable energy.
FAQ section:
How does methanol compare to ethanol in terms of molecular weight?
While methanol has a molecular weight of 32.04 g/mol, ethanol’s molecular weight is 46.07 g/mol. This indicates that ethanol is heavier than methanol, which affects their properties and uses. Methanol’s lighter weight and higher toxicity make it more hazardous in applications compared to ethanol.
What safety measures should be in place when handling methanol?
Handling methanol requires stringent safety protocols due to its high toxicity and flammability. Professionals should use appropriate personal protective equipment (PPE) such as gloves, goggles, and respirators. Ensuring proper ventilation in working areas and using methanol only in approved, well-labeled containers are crucial for safety. Additionally, methanol spills should be promptly cleaned using methods that prevent vapor inhalation.
In conclusion, the methanol molecular weight and its associated chemical properties underscore its importance in various industrial applications. Its role in chemical synthesis and energy production highlights the significance of this compound in modern science and industry. With proper handling and understanding, methanol continues to offer significant benefits across multiple sectors, underlining the need for a careful, knowledgeable approach in its utilization.