Understanding the Measuring Unit of Energy is crucial for both everyday applications and advanced scientific research. The most commonly used unit for energy is the Joule, but grasping how this unit translates into practical measurements is essential for accurate application in fields such as physics, engineering, and even daily life. This article delves into the nuances of measuring energy, offering expert insights, real-world examples, and clear, authoritative guidance.
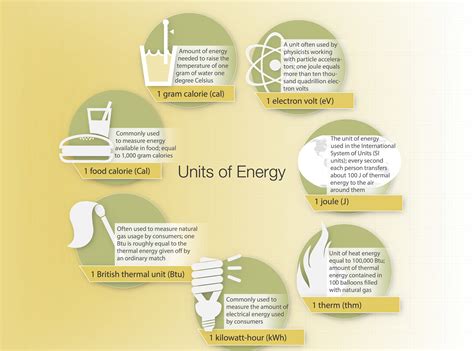
Energy, fundamentally, is the capacity to perform work or produce heat. In practical terms, it’s essential to quantify this abstract concept in a relatable manner. The Joule serves as the standard unit of energy in the International System of Units (SI), defined as the amount of energy transferred when a force of one newton is applied over a distance of one meter. Though abstract, the real-world implications of measuring energy in Joules are vast and indispensable.
Key Insights
- Understanding energy measurement in Joules is essential for scientific accuracy and practical applications.
- The Joule ties directly into physical laws, such as kinetic and potential energy calculations.
- Practical application of the Joule can enhance performance in engineering, physics, and daily life activities.
The Science Behind Joules
To truly appreciate the Joule, one must grasp its scientific foundation. At its core, the Joule measures the energy involved in physical interactions. For instance, consider kinetic energy, which is given by the formula E = 0.5mv2, where m stands for mass and v for velocity. Here, the energy is expressed in Joules, making it a direct and quantifiable aspect of any object’s motion.
Energy in Practical Scenarios
While the theoretical aspects are vital, the practical application of the Joule is even more impactful. Energy measurement in everyday life spans a variety of contexts. In engineering, for example, accurately measuring energy usage is critical in designing efficient machinery and systems. In a real-world example, consider a hydroelectric power plant. The energy generated is calculated in terms of Joules and subsequently converted into kilowatt-hours for practical utility purposes. This allows for precise quantification and optimization of energy output.
Is the Joule a small unit of energy?
While the Joule is the standard SI unit, for larger energy scales, it is often practical to use kilojoules (kJ) or megajoules (MJ). These prefixes help simplify large energy values into more manageable numbers.
How do we convert energy from Joules to other units?
Energy can be converted between units using conversion factors. One calorie, for instance, is approximately equal to 4.184 Joules. These conversions help in applying energy measurements across different scientific and practical disciplines.
Grasping the intricacies of the Joule as a unit of energy extends far beyond academic theory; it provides a solid foundation for both scientific rigor and everyday utility. By understanding and leveraging the concept of energy measurement in Joules, professionals and individuals alike can achieve greater accuracy and efficiency in various domains. This understanding enables precise calculations, optimizes performance, and fosters innovation in multiple fields.