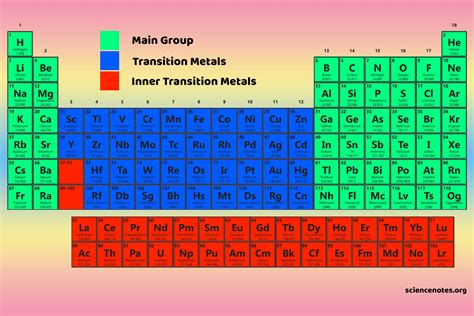
Plunging into the realm of chemistry, we often encounter a diverse spectrum of elements, each with unique properties and applications. Among these, main group elements stand out for their significance in both natural and synthetic chemistry. These elements, occupying the s and p blocks of the periodic table, serve as the foundation for a myriad of chemical reactions, materials, and biological processes. This article will explore the nuances of main group elements, revealing practical insights and actionable strategies for leveraging their potential.
Key Insights
- Main group elements are pivotal for various industrial applications.
- Understanding their electronic configurations aids in predicting reactivity.
- Strategic utilization of main group elements can optimize chemical processes.
The Chemistry of Reactivity
Main group elements are characterized by their outermost electrons in the s or p orbitals, which define their chemical behavior. The reactivity of these elements is influenced by their position on the periodic table. For instance, alkali metals in group 1 are highly reactive, given their tendency to lose one electron easily, forming +1 ions. Conversely, halogens in group 17, with their strong affinity for an electron, are also highly reactive but for different reasons. These properties are not just academic; they have practical implications in industries such as pharmaceuticals, agriculture, and manufacturing.For example, the use of lithium, a group 1 element, in rechargeable batteries showcases its reactive nature leveraged in technology. Its ability to form stable lithium ions facilitates efficient energy storage, a critical component in the advancement of renewable energy technologies.
Industrial Applications and Innovations
Beyond their inherent reactivity, main group elements contribute significantly to innovation in various sectors. Silicon, a quintessential group 14 element, epitomizes this through its ubiquitous role in electronics and computing. Silicon’s ability to form four covalent bonds allows for the creation of intricate semiconductor devices, essential for modern computing technology.Additionally, phosphorus, another group 15 element, plays a crucial role in fertilizers, which are fundamental to agricultural productivity. The utilization of phosphorus compounds in fertilizers helps meet the global demand for food, illustrating the practical applications of main group elements in everyday life.
What are the environmental considerations for main group elements?
Main group elements often have significant environmental impacts. For instance, the mining of elements like cadmium and lead raises concerns about toxicity and pollution. Therefore, it's vital to employ sustainable mining practices and recycling initiatives to mitigate these effects.
Moreover, the versatility of elements such as nitrogen and oxygen, key group 15 and 16 elements respectively, is evident in their roles in producing explosives, fertilizers, and even in atmospheric chemistry. The environmental impact, however, calls for careful handling and recycling to prevent contamination and health hazards.
How can we harness main group elements more sustainably?
Harnessing main group elements sustainably involves advanced recycling methods and the development of greener alternatives. For example, replacing traditional lead-acid batteries with lithium-ion technology can reduce environmental impact while maintaining performance.
In conclusion, main group elements, with their distinct and diverse chemical properties, are indispensable to modern science and industry. Understanding their unique behaviors and applications allows for more efficient and sustainable use, driving innovation across multiple sectors.