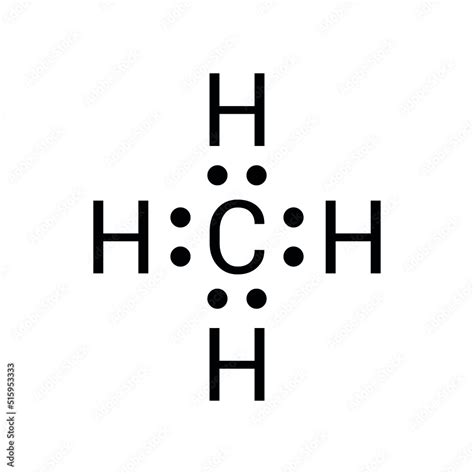
Methane, or CH4, represents a fundamental compound in organic chemistry and serves as an essential building block for understanding molecular geometry and bonding. This article delves into the intricacies of the CH4 Lewis dot structure, emphasizing the practical application of these principles for chemistry students and professionals alike.
The Lewis dot structure of methane illustrates the distribution of valence electrons in the molecule, offering a clear picture of its bonding and electron configuration. By mastering this structure, students gain a deeper comprehension of molecular shapes and the concept of electron pairs.
Key insights box:
Key Insights
- Carbon’s tetravalency enables it to form four single bonds, leading to a tetrahedral molecular geometry.
- Understanding the electron-pair repulsion helps predict the 3D structure of CH4.
- Practicing the construction of Lewis structures is critical for grasping molecular bonding and shapes.
Understanding the Lewis dot structure begins with acknowledging the valence electrons of carbon and hydrogen. Carbon, situated in group 14 of the periodic table, possesses four valence electrons. Each hydrogen atom contributes one electron. To configure the CH4 Lewis structure, place four hydrogen atoms around the central carbon atom, ensuring each hydrogen forms a single bond with carbon.
This arrangement satisfies the octet rule for carbon while ensuring each hydrogen achieves a stable electron configuration. The result is a stable tetrahedral structure where each hydrogen atom is positioned at the vertices of a tetrahedron, with carbon at the center.
The construction of the CH4 Lewis dot structure reveals an important aspect of chemical bonding: the minimization of electron pair repulsion. According to the Valence Shell Electron Pair Repulsion (VSEPR) theory, electron pairs around a central atom arrange themselves to maximize the distance between them. In CH4, the four hydrogen atoms create a perfect tetrahedral shape, with 109.5-degree angles between them. This spatial arrangement minimizes repulsion, stabilizing the molecule.
An analysis of molecular geometry follows naturally from the CH4 Lewis dot structure. The tetrahedral geometry arises because the four electron pairs surrounding the carbon atom spread out to achieve maximum separation, minimizing repulsive forces. This configuration is essential for understanding the physical and chemical properties of methane, including its relatively low boiling point and non-polarity.
A separate yet closely related analysis examines the implications of the CH4 Lewis dot structure on chemical reactivity and bonding types. The single bonds formed between carbon and each hydrogen atom are covalent bonds, characterized by the sharing of electron pairs. These bonds are non-polar due to the near-equal electronegativity of carbon and hydrogen, resulting in a molecule with no significant dipole moment.
This non-polarity influences methane’s interactions with other substances. As a result, methane is relatively inert under normal conditions and is a significant component of natural gas, highlighting its practical applications in energy production.
FAQ section:
What does the CH4 Lewis dot structure tell us about the molecule?
The CH4 Lewis dot structure indicates that methane is a tetrahedral molecule with each hydrogen atom bonded to the central carbon atom by single covalent bonds, ensuring stability through the minimization of electron pair repulsion.
How does understanding the CH4 structure help in predicting molecular behavior?
Understanding the CH4 Lewis dot structure helps predict its tetrahedral geometry, non-polar nature, and inert chemical behavior, all of which are crucial for studying its role in various chemical reactions and physical properties.
By comprehensively understanding the CH4 Lewis dot structure, students and professionals alike can unlock a wealth of knowledge about molecular geometry, bonding, and the broader implications of this simple yet foundational molecule. This knowledge forms the bedrock of advanced studies in chemistry, ensuring students are well-equipped to tackle more complex chemical phenomena.