Understanding whether HNO3 (nitric acid) is a strong acid is a common question in the field of chemistry. This guide will delve deeply into the nuances of nitric acid, addressing user pain points with actionable advice, and exploring real-world examples to demystify its properties. We’ll cover practical applications, tips for best practices, and common mistakes to avoid. By the end, you’ll have a comprehensive understanding of nitric acid’s behavior in various contexts.
Is HNO3 a Strong Acid? Clear Truth Uncovered
Nitric acid, or HNO3, is commonly encountered in laboratories and industrial settings due to its strong oxidizing properties and utility in various chemical reactions. But, is it a strong acid? This guide will uncover the truth by dissecting its chemical behavior, comparing it to other acids, and explaining its characteristics through practical examples and actionable advice.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Always neutralize nitric acid spills with sodium bicarbonate to minimize environmental impact.
- Essential tip with step-by-step guidance: Dilute nitric acid using water slowly along the jar wall to prevent exothermic reactions.
- Common mistake to avoid with solution: Misclassifying HNO3 as a weak acid due to confusion with its oxidizing properties.
It's essential to understand nitric acid’s strengths and weaknesses to use it safely and effectively.
Characteristics of HNO3: Is it a Strong Acid?
To determine if nitric acid is a strong acid, we must first understand what makes an acid 'strong'. A strong acid is one that completely dissociates in water, releasing a proton (H+) and resulting in a high concentration of hydronium ions (H3O+). Nitric acid fits this definition perfectly because it completely dissociates in aqueous solutions. This complete dissociation means that nitric acid behaves predictably in reactions and has consistent, strong acidic properties.
Let’s break down its behavior further:
- Fully Dissociaties: In water, HNO3 releases all of its protons, leading to a highly acidic solution.
- High Ionization Constants: Its high pKa value (around -1.3) reinforces its complete dissociation.
- Reactivity with Bases: It neutralizes bases efficiently due to its complete ionization, leaving no residual HNO3.
Detailed Exploration of HNO3’s Strength as an Acid
Delving deeper into nitric acid's strength involves understanding its molecular structure and behavior in aqueous solutions.
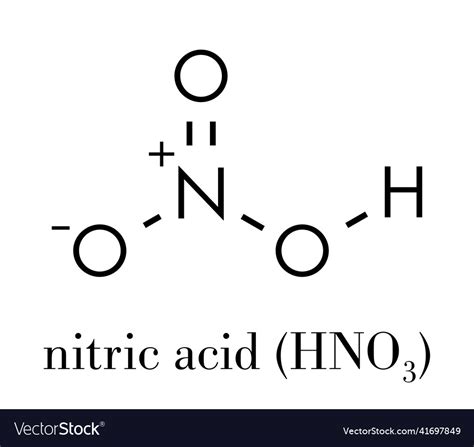
Nitric acid is a binary compound composed of hydrogen (H), nitrogen (N), and oxygen (O). Its formula is HNO3, which indicates that each molecule of nitric acid can donate one proton (H+) to a solution.
When nitric acid dissolves in water, it undergoes complete dissociation:
HNO3 → H+ + NO3-
This complete dissociation means there is no residual HNO3 left in the solution. This high degree of dissociation is what makes nitric acid a strong acid.
Another way to assess the strength of an acid is by looking at its equilibrium constant (Ka). For nitric acid:
Ka(HNO3) → Infinitely large.
A very large Ka value indicates that nitric acid completely dissociates in water, further reinforcing its classification as a strong acid.
Real-World Examples of HNO3 Usage
Understanding nitric acid's application in practical scenarios can provide more insight into its strength and behavior:
- Laboratory Use: Nitric acid is a staple in labs for metal etching, making nitrates, and oxidizing reactions. Its strength ensures it effectively removes metal oxides and oxidizes organic compounds.
- Industrial Applications: In the production of fertilizers, explosives, and dyes, nitric acid’s strong acidic and oxidizing properties are leveraged to facilitate chemical transformations.
For example, in the production of ammonium nitrate (NH4NO3) for fertilizers, nitric acid reacts with ammonia:
2NH3 + HNO3 → NH4NO3
Here, the complete dissociation of nitric acid ensures a high yield of ammonium nitrate.
Practical Tips for Handling Nitric Acid
Knowing how to handle nitric acid safely is crucial due to its strong acidic and oxidizing nature. Here are some practical tips:
- Protective Gear: Always wear appropriate protective gear including gloves, goggles, and lab coats when handling nitric acid to prevent skin and eye damage.
- Proper Storage: Store nitric acid in tightly sealed, corrosion-resistant containers, away from incompatible substances like flammable materials.
- Spill Management: In case of a spill, neutralize with a weak base like sodium bicarbonate and clean up immediately using appropriate protective gear.
Best Practices in Using Nitric Acid
Adhering to best practices ensures both effective use and safety when handling nitric acid:
- Gradual Dilution: Always dilute nitric acid by slowly adding it to water, never the other way around, to prevent exothermic reactions that can cause splashing.
- Avoid Mixing with Other Chemicals: Avoid combining nitric acid with incompatible substances like chlorinated substances which may result in hazardous reactions.
- Labeling: Clearly label containers of nitric acid with hazard symbols and warnings to avoid accidental misuse.
Common Mistakes to Avoid
Even seasoned chemists can make errors with nitric acid handling:
- Misclassification: Don't mistake HNO3's strong acidic properties for its oxidizing behavior. Remember, its strength lies in complete dissociation, not just its ability to oxidize.
- Improper Dilution: Avoid diluting nitric acid by adding water to it rapidly; this can cause a vigorous exothermic reaction.
- Neglecting Safety Measures: Always follow all safety protocols and avoid handling nitric acid without appropriate protective gear.
Practical FAQ
What are the safety precautions when handling HNO3?
When handling nitric acid (HNO3), safety precautions are critical due to its strong acidic and oxidizing properties. Here’s a detailed guide:
- Protective Gear: Wear gloves, goggles, and lab coats to protect your skin and eyes.
- Proper Ventilation: Work in a well-ventilated area to avoid inhaling any fumes.
- Dilution Technique: Always add nitric acid slowly to water along the jar wall to avoid exothermic reactions that can cause splashing.
- Neutralizing Spills: In case of spills, neutralize with a weak base like sodium bicarbonate and clean up with proper disposal.
- Storage: Store in tightly sealed, corrosion-resistant containers away from incompatible substances like flammable materials.
Following these steps ensures safe handling of nitric acid and prevents accidents.
How can I neutralize a nitric acid spill?
Neutralizing a nitric acid spill is crucial to minimize environmental and health hazards. Here’s how to do it safely:
- Stop the Spread: Contain the spill using absorbent materials like vermiculite or sand.
- Wear Protective Gear: Put on gloves, goggles, and lab coats before handling the spill.
- Neutralize: Use a weak base like sodium bicarbonate (baking soda) to neutralize the acid. Apply it liberally to the spill area. This will produce fizzing as the neutralization reaction occurs.
- Scrub: Once neutralized, use a brush to scrub the area to remove any remaining residue. <