The chemical compound carbon tetrachloride (CCl4) has long been a subject of interest in the field of chemistry, primarily due to its unique properties and diverse applications. Carbon tetrachloride is employed in various industrial processes, including as a solvent, in refrigerants, and historically in fire extinguishers. Understanding whether CCl4 is polar or nonpolar requires a deep dive into its molecular structure and the nature of its chemical bonds.
Molecular Structure of CCl4
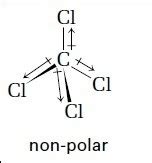
CCl4 exhibits a tetrahedral molecular geometry, with the carbon atom positioned at the center and four chlorine atoms equally spaced around it. This spatial arrangement ensures that the dipole moments created by the electronegativity difference between carbon and chlorine do not cancel out. Despite the polar nature of the C-Cl bonds, the symmetrical geometry of the molecule results in a net zero dipole moment, leading to its classification as a nonpolar molecule. In essence, the vector sum of the dipole moments points in opposite directions, effectively neutralizing each other.
Impact of Symmetry on Polarity
The symmetrical tetrahedral shape of CCl4 is pivotal in determining its nonpolarity. This symmetry ensures that the positive and negative charges are balanced, resulting in a molecule that lacks a net dipole moment. A nonpolar molecule like CCl4 does not readily dissolve in polar solvents like water, but rather aligns well with other nonpolar substances. This characteristic is critical in numerous applications where solvent properties are crucial. For instance, in industrial applications, CCl4’s nonpolar nature makes it an excellent solvent for substances like fats and oils, which are also nonpolar.
Key Insights
- Molecular Geometry: The tetrahedral geometry of CCl4 is critical in rendering the molecule nonpolar.
- Symmetry and Polarity: Despite the polar C-Cl bonds, the molecule's symmetrical shape leads to a net nonpolar molecule.
- Industrial Applications: Its nonpolar nature makes CCl4 suitable for dissolving nonpolar substances like fats and oils.
Why is CCl4 considered nonpolar despite having polar bonds?
CCl4 is nonpolar due to its symmetrical tetrahedral geometry, which cancels out the dipole moments of the C-Cl bonds, leading to a net zero dipole moment.
Does the Polarity of CCl4 Affect Its Solubility?
Indeed, the nonpolar nature of CCl4 significantly impacts its solubility characteristics. It is largely insoluble in polar solvents such as water but dissolves well in nonpolar solvents like hexane or chloroform. This behavior is grounded in the principle of “like dissolves like,” which is fundamental in chemical solubility. Because CCl4 interacts favorably with other nonpolar substances, it finds extensive use in extraction processes where separating nonpolar compounds is necessary. Additionally, this solubility property is critical in analytical chemistry for purifying substances based on polarity differences.
Environmental and Health Implications of CCl4
While CCl4’s nonpolar nature and symmetry make it beneficial in many industrial applications, its environmental and health implications cannot be overlooked. Due to its persistence in the environment and potential to cause liver damage and other health issues, the usage of CCl4 has significantly declined over the years. It is now largely replaced by safer alternatives in most applications, underscoring the balance between chemical utility and environmental stewardship. Understanding the polarity and behavior of CCl4 remains crucial for developing safer and more sustainable industrial practices.
The molecular intricacies of CCl4 underline the importance of symmetry in determining a molecule’s polarity. Its nonpolar nature is not just a chemical curiosity but a foundational principle in numerous practical and industrial applications. As we continue to explore and develop new chemical entities, the insights gleaned from compounds like CCl4 will guide us towards more responsible and effective chemical practices.