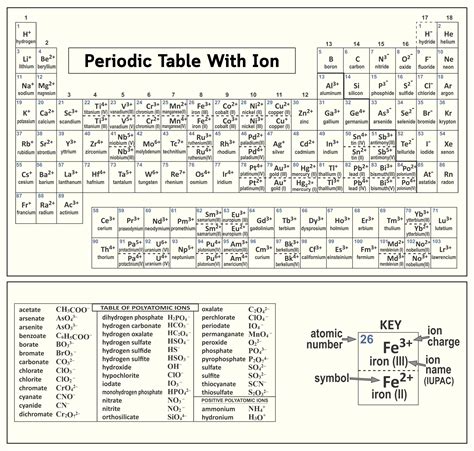
Understanding the ion periodic table is essential for any chemistry student or professional. With its rich history and evolving relevance in modern science, the periodic table continues to be a pivotal tool in the study of elements and their unique properties. Delving into the nuances of ions, especially within the context of the periodic table, offers insights into their stability, charge, and reactivity, forming a strong foundation for advanced chemical research and applications.
Key insights box:
Key Insights
- Primary insight with practical relevance: Ions, either cations or anions, are integral to understanding chemical bonding and reactivity, particularly in the context of their placement on the periodic table.
- Technical consideration with clear application: Transition metals, for example, exhibit complex ionic states due to their ability to lose and gain electrons in multiple ways, impacting their use in catalysts and alloys.
- Actionable recommendation: For students and professionals, mastering the concept of electron configuration and ionization energies is critical for predicting and manipulating ionic behavior in various chemical reactions.
Ion formation and the periodic table: An exploration Ions play a crucial role in chemistry, representing atoms or molecules that have a net electric charge due to the loss or gain of one or more electrons. Their formation is directly influenced by an element’s position on the periodic table. For instance, elements on the right side of the table tend to gain electrons to achieve a stable noble gas configuration, forming anions. Conversely, elements on the left are more likely to lose electrons, resulting in the formation of cations. Understanding this relationship between ionization and placement on the periodic table is fundamental for predicting how elements will behave in chemical reactions.
Transition metals and ionic states: A closer look Transition metals, found in the d-block of the periodic table, demonstrate unique ionic characteristics that set them apart from main group elements. Due to their partially filled d orbitals, transition metals can exhibit a range of oxidation states, making them incredibly versatile in catalytic and material science applications. For example, iron can exist in multiple ionic states (+2, +3, etc.), each of which has distinct chemical properties and uses. This complexity requires a detailed understanding of their electron configurations and ionization energies to fully exploit their potential in various industrial processes.
FAQ section:
What is the significance of ions in chemical reactions?
Ions are significant in chemical reactions because they determine the reactivity and bonding characteristics of elements. Understanding the nature of ions helps predict how different elements will interact and form compounds, thus aiding in the synthesis of new materials and the understanding of reaction mechanisms.
How do ionization energies affect ionic formation?
Ionization energy, the energy required to remove an electron from an atom, directly impacts the likelihood of an element to form ions. Elements with low ionization energies are more likely to form cations, whereas those with high electron affinities are inclined to form anions. This understanding is vital for predicting the behavior of elements in various chemical environments.
The periodic table, enriched with the knowledge of ions, becomes an invaluable resource for chemists and scientists alike. By focusing on the practical aspects of ion formation and their roles in chemical processes, professionals and students can advance their comprehension and application of chemical principles in both academic and industrial contexts.