Mastering the IF5 Lewis Structure: Uncover Key Insights and Chemistry Secrets
Understanding the IF5 Lewis structure is crucial for any chemistry enthusiast, whether you’re a student tackling advanced organic chemistry or a professional scientist diving into interhalogen compounds. The IF5 molecule can be challenging, but with a clear and methodical approach, you’ll unlock its secrets and grasp its complexity. In this guide, we’ll provide a step-by-step roadmap to master the IF5 Lewis structure, along with practical examples and actionable advice.
Problem-Solution Opening: Conquering the IF5 Lewis Structure
Many chemistry students struggle with drawing and understanding the Lewis structures of complex molecules like IF5. The process involves multiple layers of understanding, from determining the central atom and valence electrons to accounting for bonding and lone pairs. This often leads to confusion, mistakes, and frustration. To tackle this problem head-on, this guide will provide a clear, problem-solving approach with actionable advice, ensuring you can confidently draw the IF5 Lewis structure and understand its underlying chemistry.
Quick Reference
Quick Reference
- Immediate action item: Sketch a preliminary Lewis structure.
- Essential tip: Use VSEPR theory to predict the molecular geometry.
- Common mistake to avoid: Miscounting valence electrons.
Detailed How-To Section: Drawing the IF5 Lewis Structure
Drawing the IF5 Lewis structure requires careful consideration of the molecule’s components and their interactions. Follow these steps:
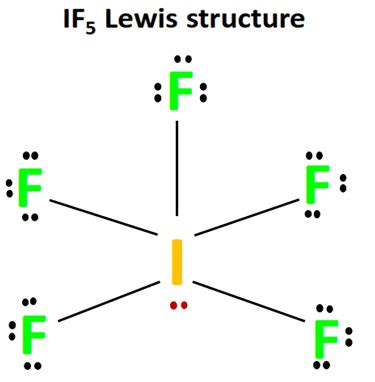
Step 1: Determine the total number of valence electrons.
Iodine (I) is in Group 17 and has 7 valence electrons. Fluorine (F) also has 7 valence electrons, and since there are five fluorine atoms, this totals to 42 electrons from fluorine. Add the 7 from iodine, giving you 49 valence electrons.
Step 2: Identify the central atom.
In IF5, iodine is the central atom. This choice is based on iodine’s lower electronegativity compared to fluorine and its ability to accommodate more than eight electrons due to its availability of d-orbitals.
Step 3: Distribute the valence electrons.
Place one electron pair between each I-F bond to form five single bonds. This accounts for 10 electrons. With 39 electrons left, place the remaining electrons on the central iodine atom, starting with lone pairs.
Step 4: Complete the octet of the central atom.
To complete the octet on the central iodine atom and considering the d-orbitals, place three lone pairs on iodine, using up 6 electrons. Now, iodine will have 10 electrons around it, which is acceptable given its ability to expand its octet.
Step 5: Check the structure for formal charges.
Ensure the structure adheres to the octet rule for all atoms. In IF5, each fluorine will have 8 electrons (including one bond pair and three lone pairs), and iodine will have an expanded octet.
This step-by-step process is fundamental to accurately representing the IF5 Lewis structure.
Detailed How-To Section: Understanding IF5 Geometry and Hybridization
Beyond drawing the Lewis structure, understanding the geometry and hybridization of IF5 is crucial. Follow these steps:
Step 1: Use VSEPR theory to predict molecular geometry.
IF5 has a total of five bonding pairs and one lone pair around the central iodine atom, which sums up to six electron groups. According to VSEPR theory, these will arrange themselves to minimize repulsion, resulting in a square pyramidal geometry.
Step 2: Determine the hybridization state of the central atom.
For iodine in IF5, the hybridization must accommodate six electron groups. This requires the involvement of iodine’s 5p and one 5d orbitals, leading to sp3d2 hybridization.
Step 3: Confirm the structure with molecular modeling.
Use a molecular model kit or online tool to visualize the predicted geometry and hybridization. This can help cement your understanding and verify the theoretical predictions.
These steps will guide you through the critical concepts needed to fully understand the IF5 molecule.
Practical FAQ Section
Why is the IF5 Lewis structure more complex than typical molecules?
The complexity of IF5 arises from iodine’s ability to use its d-orbitals to accommodate more than eight electrons. Unlike carbon, which typically forms four bonds, iodine in IF5 forms five bonds and has three lone pairs, leading to its unique square pyramidal shape and sp3d2 hybridization.
What common errors should I avoid when drawing the IF5 Lewis structure?
Common errors include miscounting valence electrons, incorrectly determining the central atom, or misunderstanding iodine’s capability to expand its octet. Double-check your calculations, ensure the correct central atom, and remember that iodine’s d-orbitals play a critical role in forming more than eight bonds.
How can I verify my IF5 Lewis structure?
To verify your structure, use a molecular model kit to physically build the molecule, ensuring the correct connectivity and geometry. Alternatively, use online molecular modeling software like Avogadro or Jmol. These tools allow you to visualize and check your structure’s geometry and hybridization.
By following this comprehensive guide, you’ll gain a deep understanding of the IF5 Lewis structure, the geometry, and hybridization of the molecule. You’ll move from confusion to confidence with actionable steps and practical solutions, ensuring you can tackle complex molecules with ease.