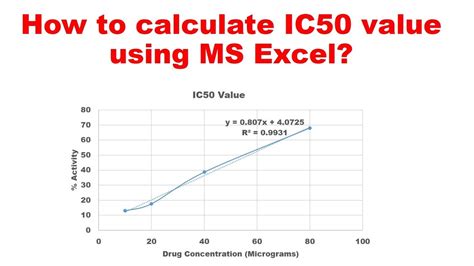
The IC 50, a critical metric in bioassays, signifies the concentration of a compound required to achieve 50% of its maximum effect. Understanding and mastering this parameter is paramount for researchers in pharmaceuticals, pharmacology, and environmental sciences. This article delves into the secrets of optimizing the IC 50, backed by evidence-based insights and practical applications.
Key Insights
- Primary insight with practical relevance: Achieving a lower IC 50 indicates higher potency and efficacy, which is crucial for drug development.
- Technical consideration with clear application: Careful execution of experimental design, including proper control setup and statistical analysis, is fundamental to obtaining accurate IC 50 values.
- Actionable recommendation: Implement standard operating procedures (SOPs) for replicability and accuracy in bioassay experiments.
Understanding the Importance of IC 50 in Drug Development
The IC 50 serves as a primary parameter in assessing drug efficacy. When developing new pharmaceutical compounds, determining an accurate IC 50 value provides a reliable indicator of potency. A lower IC 50 implies that a smaller concentration of the compound can produce the desired effect, indicating a more potent drug. This is not just theoretical—it has direct implications on the cost, safety, and therapeutic index of the drug. Accurate IC 50 determination allows for the fine-tuning of dosages, ensuring optimal therapeutic outcomes while minimizing side effects.Techniques for Accurate Determination of IC 50
Accurate measurement of the IC 50 relies heavily on robust experimental design and execution. Researchers should employ a well-planned series of concentration-response curves, measuring responses at various concentrations. A key technical consideration is the inclusion of appropriate controls: untreated cells and positive controls to ensure the assay’s reliability. Statistical analysis methods, such as curve fitting and regression models, play a critical role in deriving the IC 50 values. Advanced software tools can facilitate the analysis, allowing for the precise determination of the IC 50 even from small datasets.What is the difference between IC 50 and EC 50?
IC 50 refers to the inhibitory concentration for 50% effect, commonly used in cytotoxic assays. EC 50, on the other hand, denotes the effective concentration for 50% effect and is widely used in assays assessing biological effects such as receptor binding or enzyme inhibition. While both metrics aim to gauge the potency of a compound, they differ in context and application.
How does the IC 50 value affect drug approval?
The IC 50 value is a critical parameter in the drug approval process as it reflects the efficacy of a drug. Regulatory agencies require comprehensive data on the drug’s potency and safety, and the IC 50 value provides a quantifiable measure of the drug’s effectiveness. It helps in determining the therapeutic dose and can influence the drug's overall safety profile.
Understanding and mastering the IC 50 is pivotal for advancing scientific research and developing effective therapeutics. Researchers must focus on meticulous experimental design and rigorous data analysis to ensure accurate IC 50 determinations. By integrating these proven strategies, researchers can enhance their bioassay results, contributing to the development of more potent and safer drugs.