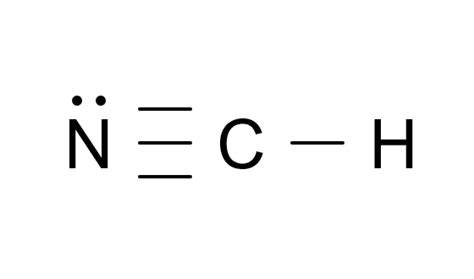Hydrogen cyanide (HCN) is a vital compound in chemistry, especially for those delving into organic chemistry or those studying industrial applications of chemical compounds. As with many chemical structures, understanding the Lewis structure of hydrogen cyanide offers a gateway into comprehending its reactivity, properties, and implications. This guide will break down the process in a practical, easy-to-follow manner, complete with actionable advice and clear examples.
Understanding Hydrogen Cyanide: The Why and the How
Hydrogen cyanide (HCN) is a simple molecule but packs a punch in terms of reactivity and applications. From being a key player in the synthesis of plastics to serving as an intermediate in pharmaceutical manufacturing, the correct understanding of HCN’s structure can vastly improve your grasp of chemical bonding and molecular interactions. This guide is designed to walk you through the foundational steps of drawing the Lewis structure of HCN, ensuring that you not only understand the ‘what’ but also the ‘why’ behind each step.
Quick Reference
Quick Reference
- Immediate action item: Count valence electrons to ensure balance in Lewis structure
- Essential tip: Use the octet rule as a guide to understand electron distribution
- Common mistake to avoid: Forgetting to place lone pairs appropriately to fulfill the octet rule
Detailed How-To: Drawing the Lewis Structure of HCN
Drawing the Lewis structure of hydrogen cyanide may seem daunting, but breaking it down step-by-step makes it manageable. Let’s start by understanding the atomic configuration and move through each stage with clarity.
Step 1: Counting Valence Electrons
Each atom in a molecule contributes a number of valence electrons. Hydrogen (H) has 1 valence electron and carbon © has 4, while nitrogen (N) has 5. For HCN:
Total valence electrons: 1 (H) + 4 © + 5 (N) = 10 electrons
Step 2: Placing the Central Atom
Carbon, being the most electronegative among H, C, and N in this context, will be the central atom. Hydrogen, the least electronegative, will be on the outside.
Set up: C between H and N
Step 4: Completing the Lewis Structure
After placing hydrogen on one end and nitrogen on the other, ensure all atoms reach their octet where possible:
- Hydrogen reaches its duet (2 electrons) with 1 electron from Carbon.
- Carbon forms a triple bond with Nitrogen to fulfill its octet.
- Nitrogen, having 5 valence electrons, shares three pairs with Carbon, reaching its octet.
Step 5: Verifying the Structure
Finally, check to ensure all atoms are satisfied with their electron count:
- Hydrogen: 2 electrons (full duet)
- Carbon: 8 electrons (triple bond with Nitrogen)
- Nitrogen: 8 electrons (triple bond with Carbon)
Practical Application: Why the Lewis Structure Matters
Understanding the Lewis structure of hydrogen cyanide is not just an academic exercise. It’s a practical tool for predicting the molecule’s behavior in reactions. For instance:
- Chemical reactivity: The triple bond between carbon and nitrogen in HCN makes it highly reactive, especially in substitution and addition reactions.
- Industrial uses: HCN is often used in the production of plastics like acrylic fibers and in the pharmaceutical industry for synthesizing various compounds.
FAQ: Answering Your Most Common Questions
Why is the triple bond in HCN important?
The triple bond in HCN significantly affects its reactivity. This bond makes HCN a potent molecule in various chemical reactions, including its role in nitrile synthesis, where it serves as a building block for creating a wide range of chemical compounds.
How does the Lewis structure of HCN relate to its toxicity?
Though the Lewis structure doesn’t directly show toxicity, understanding the molecule’s reactivity and stability provides insight into its harmful effects. HCN is known for its high toxicity due to its ability to inhibit cellular respiration by binding to cytochrome c oxidase, an essential protein in the electron transport chain.
This guide aims to demystify the Lewis structure of hydrogen cyanide by walking through each step clearly and providing real-world context. Whether you are a student, professional chemist, or simply curious, understanding this structure will empower you with knowledge that extends far beyond the classroom. Remember, the key is to practice, verify, and apply these principles to both academic and practical settings.