To delve into the process of finding mass using volume and density is to explore a fundamental principle in physics and engineering that bridges theoretical concepts with practical application. This principle, governed by the equation mass = density × volume, holds a pivotal place in numerous fields, from material science to aerospace engineering. Whether you are dealing with the precise calculations required for chemical reactions or assessing the load-bearing capacity of construction materials, understanding this principle can provide critical insights.
Key insights box:
Key Insights
- Primary insight with practical relevance: Knowing the density and volume of a substance allows you to determine its mass, which is crucial in various scientific and engineering applications.
- Technical consideration with clear application: Precision in calculations is essential; even minor errors in measuring density or volume can lead to significant discrepancies in mass.
- Actionable recommendation: Always double-check your measurements and calculations to ensure accuracy when determining mass.
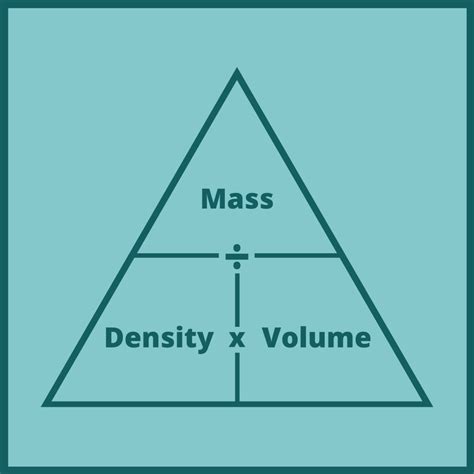
Understanding how to find mass from volume and density requires a solid grasp of the formula: mass (m) equals density (ρ) multiplied by volume (V). This formula can be written as:
m = ρ × V
This relationship is grounded in the definition of density, which is the mass per unit volume of a substance. For instance, if you know that the density of water is approximately 1 gram per cubic centimeter (g/cm³), and you have a container with a volume of 5 liters (which is equivalent to 5,000 cm³), you can calculate the mass of the water by multiplying the density by the volume:
m = 1 g/cm³ × 5,000 cm³ = 5,000 grams
This straightforward calculation highlights the importance of accurate measurements and unit conversions in practical scenarios.
In practical applications, determining mass through volume and density is crucial in fields such as materials science and engineering. For example, when designing a structure, engineers must calculate the mass of materials used to ensure that structural elements can support the expected loads without failure. Using the formula, they can determine the mass of steel required for a beam by measuring its volume and knowing its density.
Another key area where this principle is applied is in pharmaceutical sciences. When formulating medications, precise mass calculations ensure that the correct dosages are mixed with the proper solvents, which depends on accurate density and volume measurements.
Another analysis area involves environmental science, where understanding the density and volume of pollutants in air or water helps in assessing the mass of contaminants present in a given volume. For instance, determining the mass of a pollutant like lead in water requires knowing its density and the volume of the water sample, which is essential for regulatory compliance and cleanup efforts.
FAQ section:
How does precision affect the accuracy of mass calculations?
Precision in measuring density and volume is critical because any errors can lead to significant discrepancies in the calculated mass. For example, if a volume measurement is slightly off by just a few cubic centimeters, this can result in a noticeable error in mass, especially when dealing with large volumes.
What unit conversions are necessary when using this formula?
Unit consistency is essential when using the formula. Typically, volume should be in cubic units (like cm³, m³) and density in mass per cubic unit (like g/cm³, kg/m³). Converting between different units, such as liters to cubic centimeters, is necessary to maintain consistency and ensure accurate calculations.
In conclusion, the principle of calculating mass from volume and density is a cornerstone in various scientific and engineering domains. It requires precise measurements and a clear understanding of the fundamental formula. Whether you’re dealing with water in a chemical reaction, structural materials in engineering, or pollutants in environmental science, accurate mass determination is indispensable for successful outcomes.