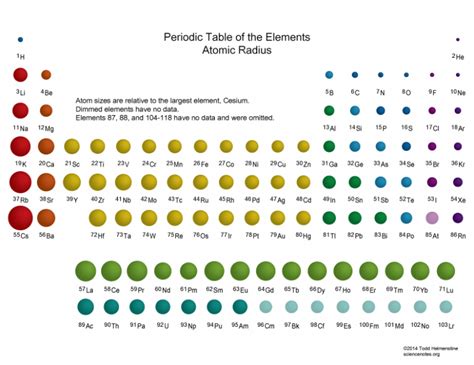
The atomic radius of an element is a critical aspect in understanding its chemical and physical properties. It represents the size of an atom, which directly influences how it bonds with other atoms, its reactivity, and its overall behavior in various environments. This quick, simple guide aims to provide clarity on atomic radius, its measurement, and its significance in chemistry.
Key insights box:
Key Insights
- Atomic radius is essential for predicting an element’s chemical reactivity and bonding behavior.
- The trend in atomic radius is primarily influenced by the number of electron shells and the effective nuclear charge.
- Understanding atomic radius helps in designing new materials with specific properties.
In the periodic table, atomic radius exhibits a distinct trend. Generally, the atomic radius increases as we move down a group and decreases across a period from left to right. This behavior can be explained by the arrangement of electrons in shells around the nucleus. When moving down a group, new electron shells are added, thus increasing the distance between the nucleus and the outermost electrons, leading to an increase in atomic radius. Conversely, as we move across a period, the number of protons in the nucleus increases, leading to a greater effective nuclear charge that pulls the electrons closer, thereby decreasing the atomic radius.
Understanding the trends in atomic radius is crucial for predicting chemical reactivity. Larger atoms, typically those with a greater atomic radius, tend to be less reactive because their outer electrons are further from the nucleus and are less tightly held. These electrons are available to participate in chemical reactions. For example, elements like potassium and sodium, which are at the bottom of Group 1, have larger atomic radii and are highly reactive due to the ease with which they lose their outermost electron to form a positive ion.
Smaller atoms, with smaller atomic radii, often exhibit higher reactivity. For instance, elements like fluorine and chlorine, located at the top right of the periodic table, have small atomic radii and high electronegativity, making them highly reactive. Their small size allows the high effective nuclear charge to draw electrons in closely, leading to strong attractions between oppositely charged particles.
Two factors primarily influence atomic radius: electron configuration and nuclear charge. As we move down a group in the periodic table, additional electron shells increase the atomic radius, making elements larger and typically less reactive. This is evident in the group of alkali metals, where elements like lithium, sodium, and potassium show a progressive increase in atomic radius, correlating with their decreasing reactivity down the group.
On the other hand, moving across a period from left to right, the increase in nuclear charge has a more significant impact than the addition of a new electron shell. More protons in the nucleus exert a stronger positive charge, pulling electrons closer to the nucleus, which decreases the atomic radius. This phenomenon can be seen in the trend from sodium to chlorine across Period 3, where atomic radius diminishes as the nuclear charge increases.
FAQ section:
What is the main reason for the variation in atomic radius across the periodic table?
The variation in atomic radius is mainly due to two factors: the number of electron shells and the effective nuclear charge. Down a group, additional electron shells increase the atomic radius, while across a period, the increase in nuclear charge pulls electrons closer to the nucleus, decreasing the atomic radius.
Why do elements with larger atomic radii tend to be less reactive?
Elements with larger atomic radii have outer electrons that are further from the nucleus and are less tightly held. These electrons are not as readily available to participate in chemical reactions, which reduces the reactivity of the element.
This guide emphasizes the importance of understanding atomic radius in predicting chemical behavior, material design, and various applications in science and technology. Mastery of these principles allows scientists and engineers to innovate, creating new compounds and materials with desired properties and reactivity levels.