Performing mole calculations from grams is a fundamental skill in chemistry that enables you to bridge the gap between the macroscopic world, which we can measure and see, and the microscopic world of atoms and molecules that are otherwise invisible. This guide will provide you with step-by-step instructions, actionable advice, real-world examples, and best practices to ensure you understand and can apply mole calculations confidently.
Introduction
Mole calculations from grams form the foundation of chemical stoichiometry, allowing you to determine the amounts of substances involved in chemical reactions. Whether you’re a student learning chemistry for the first time or a professional needing a refresher, mastering this skill is critical for understanding both theoretical and practical aspects of chemistry.
In this guide, we will walk you through the process of converting grams to moles, explore the essentials with a quick reference guide, delve into detailed examples, and address common questions that arise. By the end of this guide, you will have the knowledge to perform mole calculations with confidence.
Understanding the Problem-Solution Approach
A common problem faced by many students is the difficulty in converting grams to moles due to the complexities of the atomic mass unit (amu) and the concept of the mole itself. The mole is a fundamental unit in chemistry that allows scientists to count atoms by weighing them. Essentially, one mole of any substance contains exactly 6.022 x 1023 entities (Avogadro’s number). This guide will focus on problem-solving techniques that demystify this process, providing clear and actionable steps to transform grams into moles efficiently.
Here's how we'll tackle this problem:
- Identify the specific substance for which you need to calculate moles.
- Find the molar mass (molecular weight) of the substance from a periodic table or a reliable chemical reference.
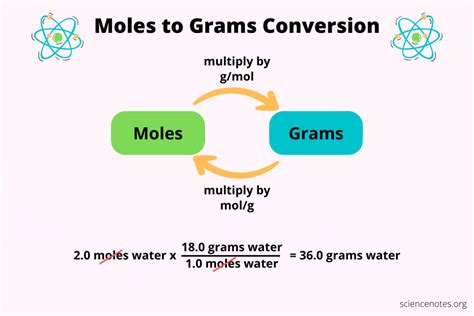
- Use the formula: moles = grams / molar mass to perform the calculation.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Use a periodic table to find the atomic or molecular mass of your substance for accurate calculations.
- Essential tip with step-by-step guidance: Always make sure your units are consistent—grams need to be divided by the molar mass in grams per mole.
- Common mistake to avoid with solution: Failing to convert grams to the correct unit when calculating moles, which leads to incorrect results.
Detailed How-To: Converting Grams to Moles
Let's walk through an example. Suppose you have 25 grams of water (H2O) and you want to determine the number of moles present. Here’s the step-by-step process:
Step 1: Identify the substance
In this case, the substance is water (H2O).
Step 2: Determine the molar mass
To determine the molar mass of water:
- Find the atomic mass of hydrogen (H): approximately 1.008 grams per mole.
- Find the atomic mass of oxygen (O): approximately 16.00 grams per mole.
- Calculate the molar mass: (2 x 1.008 g/mol) + (1 x 16.00 g/mol) = 18.016 grams per mole.
Step 3: Calculate moles
Now use the formula: moles = grams / molar mass
Substitute the values:
moles = 25 grams / 18.016 grams per mole ≈ 1.39 moles
Step 4: Interpret the result
This means that 25 grams of water (H2O) is approximately equal to 1.39 moles of water.
This method can be applied to any substance by following the same process: identify the substance, determine its molar mass, and calculate the moles using the formula.
Detailed How-To: Calculating Moles for Various Substances
Here are additional examples to illustrate the process for different substances, each using the same step-by-step approach:
Example 1: Sodium Chloride (NaCl)
Suppose you have 58.44 grams of sodium chloride:
- Identify the substance: Sodium chloride (NaCl).
- Determine the molar mass:
- Molar mass of Na = 22.99 grams per mole
- Molar mass of Cl = 35.45 grams per mole
- Total molar mass of NaCl = 22.99 g/mol + 35.45 g/mol = 58.44 grams per mole
- Calculate moles: moles = 58.44 grams / 58.44 grams per mole = 1 mole
Example 2: Glucose (C6H12O6)
Suppose you have 180 grams of glucose:
- Identify the substance: Glucose (C6H12O6).
- Determine the molar mass:
- Molar mass of C = 12.01 grams per mole (6 x 12.01 g/mol = 72.06 grams)
- Molar mass of H = 1.008 grams per mole (12 x 1.008 g/mol = 12.096 grams)
- Molar mass of O = 16.00 grams per mole (6 x 16.00 g/mol = 96.00 grams)
- Total molar mass of C6H12O6 = 72.06 g/mol + 12.096 g/mol + 96.00 g/mol = 180.156 grams per mole
- Calculate moles: moles = 180 grams / 180.156 grams per mole ≈ 0.999 moles
Practical FAQ
What if I make a mistake in finding the molar mass?
It’s common to make a mistake when determining the molar mass. To ensure accuracy:
- Double-check the atomic masses from a reliable periodic table.
- Verify the chemical formula for the compound you’re calculating.
- Break down the calculation into smaller parts to avoid errors.
For instance, if you miscalculate the molar mass of glucose as 180 grams per mole instead of 180.156 grams per mole, you’ll find:
moles = 180 grams / 180 grams per mole = 1 mole.
However, this will result in a very slight difference since the correct molar mass is slightly higher. Such precision is crucial in scientific calculations.
How do I handle very large or very small molar masses?
When dealing with very large or very small molar masses, it’s important to:
- Use scientific notation to express molar masses for better readability.
- Use a calculator for precise division, especially when dividing large numbers or very small fractions.
- Ensure that you correctly use units; grams per