Understanding valence electrons is crucial for grasping chemical bonding and the behavior of elements in various compounds. Oxygen is particularly important as it forms the backbone of many molecules we encounter daily, from the air we breathe to the water we drink. This guide will offer you step-by-step guidance with actionable advice, practical solutions, and real-world examples to help you master the concept of valence electrons, focusing on oxygen.
Introduction to Valence Electrons
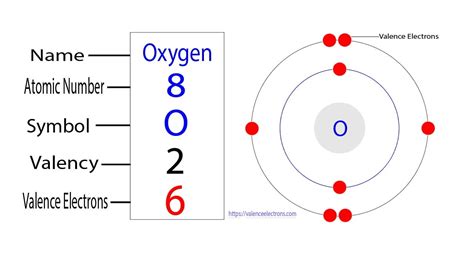
Valence electrons are the outermost electrons of an atom, and they play a key role in chemical reactions because they are involved in the formation of chemical bonds. Understanding these electrons can explain why certain elements react with each other and how molecules form. For oxygen, having six valence electrons determines its reactivity and bonding patterns, which is fundamental to comprehending its behavior in chemical compounds.
Why Valence Electrons Matter
When we delve into the chemistry of oxygen, it’s essential to grasp its electron configuration to understand its role in various chemical reactions. Oxygen’s valence electrons are crucial for forming double bonds in molecules such as water (H2O) and carbon dioxide (CO2). This guide will help you navigate through the basics of electron counting, starting from a fundamental level and building your knowledge progressively.
Here's a problem-solving approach: If you're struggling with identifying valence electrons or understanding oxygen’s role in chemical bonding, this guide will help you tackle those challenges. By the end of this, you'll have a clear understanding of oxygen’s electron configuration, how to determine its valence electrons, and practical applications.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Determine the total number of electrons in oxygen by adding its atomic number (8) and its valence electrons (6).
- Essential tip with step-by-step guidance: To find valence electrons, look at the group number in the periodic table for non-transition metals (Group 16 for oxygen). Alternatively, subtract the number of inner shell electrons from the total atomic number.
- Common mistake to avoid with solution: Mistaking core electrons for valence electrons. Ensure you count only the outermost electrons, not the inner filled shells.
Detailed Understanding of Valence Electrons
To get a detailed understanding of valence electrons, let’s first explore what electron configurations entail. Electron configurations describe how electrons are distributed in an atom’s orbitals. For oxygen, the electron configuration is written as 1s22s22p4. This indicates that oxygen has two electrons in its first shell, two in its second shell, and four in its p orbitals in the outermost shell.
Breaking this down, we see:
- 1s2: Two electrons in the 1s orbital
- 2s2: Two electrons in the 2s orbital
- 2p4: Four electrons in the 2p orbitals
From this, we identify the valence electrons by focusing on the outermost shell, which for oxygen is the second shell, containing the 2p4 electrons. Since these are the electrons furthest from the nucleus and available for chemical reactions, oxygen has six valence electrons.
Determining Valence Electrons for Oxygen
Identifying valence electrons is straightforward for oxygen. Here’s a step-by-step method to determine them:
- Start with the atomic number: Oxygen has an atomic number of 8, meaning it has 8 electrons.
- Write the electron configuration: From the atomic number, we get the electron configuration as 1s22s22p4.
- Identify the valence shell: The outermost shell, which for oxygen is the second shell, contains the 2p4 electrons.
- Count the valence electrons: Here, we have four 2p electrons and two 2s electrons in the outer shell, totaling six valence electrons.
This method ensures you don’t mistake core electrons for valence electrons, which can be a common error. Understanding this helps in predicting oxygen’s bonding capacity and reactivity.
Practical Application: Oxygen’s Role in Chemical Reactions
Now that you know oxygen has six valence electrons, let’s explore how this knowledge applies in real-world scenarios. Oxygen’s two unpaired electrons in its 2p orbital enable it to form two covalent bonds with other atoms, typically hydrogen in water or carbon in carbon dioxide. This bonding pattern is central to the stability and formation of various molecules.
For example, in the formation of water (H2O), each hydrogen atom shares a pair of electrons with the oxygen atom, fulfilling the octet rule for all atoms involved. This shared electron pair forms a covalent bond, holding the water molecule together.
FAQs on Valence Electrons and Oxygen
What is the best way to understand valence electrons?
To grasp valence electrons, start by looking at the periodic table. Non-transition metals follow group numbers to determine valence electrons. For oxygen (Group 16), simply count six electrons as valence electrons. Practice by applying this method to other elements and their compounds. Familiarize yourself with electron configurations and how to identify the outermost shell.
Why does oxygen typically form two bonds?
Oxygen has six valence electrons, and it commonly seeks to achieve a full outer shell (octet). To complete its octet, oxygen forms two covalent bonds with atoms like hydrogen, sharing two pairs of electrons. This is why in water, each oxygen atom bonds with two hydrogen atoms, fulfilling the octet rule and ensuring stability.
Can valence electrons change?
Valence electrons can change during chemical reactions as atoms gain or lose electrons to achieve a more stable configuration. In ionic compounds, atoms can lose or gain electrons to become ions, altering their valence electron count. For example, sodium (Na) loses one electron to form Na+, whereas chlorine (Cl) gains one electron to form Cl-.
This guide has taken you through the fundamental concepts of valence electrons, focusing specifically on oxygen. By understanding oxygen’s six valence electrons, you’ll be better prepared to tackle more complex chemical concepts and the behavior of molecules in various environments. Through practical examples and clear, actionable guidance, we hope this resource helps demystify the role of valence electrons and oxygen’s reactivity.