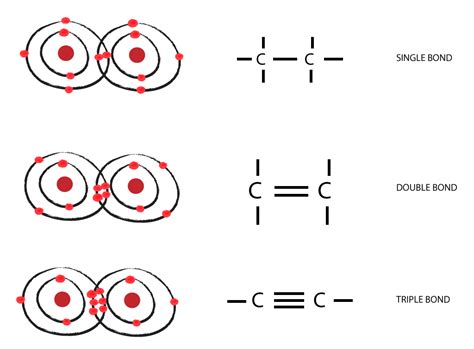
Carbon’s versatility in forming bonds is a fundamental concept in organic chemistry, crucial for understanding molecular structures and chemical reactions. With its four valence electrons, carbon can form up to four covalent bonds with other atoms, enabling it to create complex and diverse molecular architectures. This unique bonding capacity is a cornerstone in the study of organic compounds, from simple methane to complex macromolecules like DNA.
Key Insights
- Carbon's ability to form up to four covalent bonds makes it a foundational element in organic chemistry.
- This tetravalent nature allows carbon to engage in diverse bonding patterns, including single, double, and triple bonds, and forms the backbone of organic molecules.
- Understanding carbon's bonding capabilities is essential for designing and synthesizing new organic compounds.
Understanding carbon’s bonding capabilities hinges on its electronic configuration. Carbon’s four valence electrons allow it to form four covalent bonds. This tetravalency can manifest in various forms, including single, double, and triple bonds. Single bonds involve sharing one pair of electrons, double bonds involve sharing two pairs, and triple bonds involve sharing three pairs. Each bonding pattern contributes to the structural and functional diversity of organic molecules. For example, ethylene (C2H4) showcases a double bond, whereas acetylene (C2H2) features a triple bond, demonstrating the versatility of carbon in bonding arrangements.
In addition to single, double, and triple bonds, carbon’s tetravalency also facilitates the formation of complex ring structures and branching in molecules. This versatility is essential in forming the myriad structures found in living organisms. For instance, the hexagonal structure of benzene (C6H6) highlights carbon’s capacity to form stable aromatic rings. The presence of rings and branching further enhances the structural complexity and functionality of organic compounds, playing a vital role in fields ranging from pharmaceuticals to materials science.
Can carbon form more than four bonds?
Typically, carbon forms up to four covalent bonds due to its tetravalent nature. However, under certain circumstances and in specific compounds, hypervalent carbon can form five or even six bonds, expanding its traditional bonding capacity through the involvement of d-orbitals, albeit these are less common and involve complex electronic structures.
How does carbon's bonding flexibility impact organic chemistry?
Carbon's ability to form diverse bonds underpins the complexity and diversity of organic compounds. This flexibility allows for the creation of a vast array of molecules with different properties and functions, essential for the development of new drugs, materials, and understanding biological processes.
Carbon’s capacity to form four bonds is not just a chemical curiosity; it has profound implications for the development of new technologies and the understanding of natural phenomena. As research advances, the exploration of carbon’s bonding capabilities continues to unveil new possibilities in both synthetic chemistry and natural science, showcasing the element’s pivotal role in the molecular world.