Mastering the HF Lewis structure is pivotal for understanding molecular bonding and chemistry’s fundamental principles. This guide will walk you through clear, practical steps that illuminate the structure of hydrogen fluoride, a compound of significant interest in both academic and applied chemistry. With practical insights grounded in real-world examples, we aim to make the complex simple and approachable.
Understanding the Basics of Lewis Structures
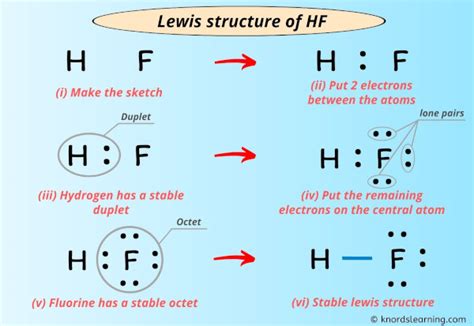
Lewis structures, introduced by Gilbert Lewis, offer a visual representation of molecules, illustrating how atoms share electrons to form covalent bonds. For hydrogen fluoride (HF), the Lewis structure helps us visualize the single covalent bond between the hydrogen and fluorine atoms. Understanding this structure is foundational for grasping more complex chemical interactions.
Key Insights
- The HF molecule comprises a hydrogen atom and a fluorine atom, showcasing a straightforward covalent bond.
- Fluorine's high electronegativity influences the electron distribution, highlighting the significance of electronegativity in bonding.
- Adhering to the octet rule is critical to achieving a stable Lewis structure.
Step-by-Step Guide to Drawing the HF Lewis Structure
Creating a Lewis structure for HF involves several systematic steps:
- Start by determining the total number of valence electrons. Hydrogen has 1 valence electron, and fluorine has 7.
- Place the least electronegative atom (hydrogen) in the center, and connect it to the more electronegative atom (fluorine) with a single bond.
- Distribute the remaining valence electrons around the outer atom (fluorine) to fulfill its octet.
- Verify the structure to ensure all atoms achieve a full octet, except hydrogen which can only hold two electrons.
Following these steps, the Lewis structure for HF illustrates a single bond and the lone pair on fluorine, underscoring the simple yet effective nature of this molecule.
Advanced Considerations in HF Bonding
When delving deeper, it’s essential to consider electronegativity differences and their impact on molecular geometry and polarity. The substantial electronegativity difference between hydrogen and fluorine results in a polar covalent bond, giving HF its dipole moment. This attribute is crucial for understanding its behavior in various chemical environments, particularly in reactions and interactions with other molecules.
Additionally, knowledge of resonance structures is vital for more complex molecules, although HF lacks the necessity for such considerations. Understanding these principles prepares you for more advanced topics, like molecular orbital theory and the nuances of quantum chemistry.
Why is the HF bond considered polar?
The HF bond is polar due to the significant electronegativity difference between hydrogen and fluorine, resulting in an uneven distribution of charge and a dipole moment.
Can HF form hydrogen bonds?
Yes, HF can form hydrogen bonds due to the presence of a highly electronegative fluorine atom, which pulls electron density away from hydrogen, creating a hydrogen bond donor.
This structured approach to understanding the HF Lewis structure offers a robust foundation for more complex molecular explorations. By mastering these basics, you equip yourself with essential knowledge to advance in your chemical studies with confidence.