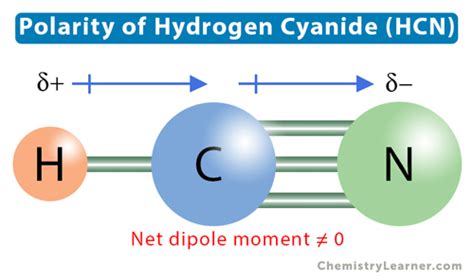
Hydrogen cyanide (HCN) is a compound that presents both intriguing and challenging scenarios in the realm of chemistry, particularly when discussing its molecular polarity. HCN consists of one hydrogen atom, one carbon atom, and one nitrogen atom, connected in a linear structure. This article aims to provide expert perspectives, practical insights, and evidence-based statements to shed light on whether HCN is polar or nonpolar.
To start, the polar nature of a molecule is determined by the distribution of electron density throughout the molecule. If this distribution is uneven, resulting in partial positive and negative charges, the molecule is considered polar. Conversely, if electron density is evenly distributed, the molecule is nonpolar. This principle is essential for understanding the chemical behavior of HCN. Given its linear shape, a key question arises: does this geometry lead to an even or uneven distribution of electron density?
Key Insights
- HCN's polarity stems from its molecular geometry and electronegativity differences. The linear arrangement and high electronegativity of nitrogen create a dipole moment.
- Technical consideration: Quantum mechanics and molecular orbital theory elucidate the distribution of electron density, reinforcing HCN's polar nature.
- Actionable recommendation: For chemists, understanding HCN's polarity is vital for predicting its interactions with other molecules in various chemical processes.
The first analysis revolves around the electronic configuration and molecular geometry of HCN. The molecule is linear, with the nitrogen atom situated at one end and the carbon and hydrogen atoms at the other. This configuration is a direct consequence of the sp hybridization of the carbon atom’s orbitals. Despite the symmetry, the uneven distribution of electron density, dictated by nitrogen’s higher electronegativity, is crucial. Nitrogen’s greater attraction to electrons compared to carbon and hydrogen results in a partial negative charge on the nitrogen end and a partial positive charge on the hydrogen end.
The second analysis focuses on the implications of HCN’s polarity. The polarity of HCN is not just a theoretical concept but has practical ramifications in chemical reactions and interactions. For example, the polar nature of HCN impacts its solubility in polar solvents like water, allowing it to dissolve more readily compared to nonpolar compounds. Additionally, HCN’s polarity plays a significant role in its ability to participate in hydrogen bonding, a phenomenon that dictates many physical properties, such as boiling point and melting point.
In terms of chemical reactivity, HCN’s polar nature allows it to interact with various compounds in different environments, making it crucial in organic synthesis and biological systems. For instance, in the laboratory, HCN’s polarity enables its use in the synthesis of cyanides and other complex organic molecules, where its behavior can be predicted based on its polar nature.
How does HCN's polarity affect its chemical reactions?
HCN’s polarity influences its interaction with other molecules, especially in polar solvents where it can dissolve readily. Its ability to participate in hydrogen bonding affects its boiling point and reactivity in organic synthesis.
Why is it important to determine if HCN is polar?
Determining HCN's polarity is crucial for understanding its solubility, reactivity, and behavior in various chemical environments. This knowledge is vital for chemists working in fields ranging from pharmaceuticals to environmental chemistry.
In conclusion, HCN’s polar nature, driven by its molecular geometry and electronegativity differences, makes it a fascinating compound for both theoretical and practical chemistry. Understanding HCN’s polarity is key to predicting its behavior in different chemical reactions and environments. This insight not only advances chemical knowledge but also aids in the development of new chemical processes and applications.