The H3O+ Lewis structure reveals a fundamental concept in chemistry, the hydronium ion, which is pivotal in understanding acidic solutions. The structure provides insight into bonding, charge distribution, and the overall molecular geometry of this ion. This article will delve into the intricacies of the H3O+ Lewis structure with an expert perspective, emphasizing practical insights and evidence-based statements enriched with real-world examples.
Key Insights
- The H3O+ ion is central to understanding Brønsted-Lowry acids.
- A detailed analysis of valence electrons is crucial for accurate representation.
- Drawing the Lewis structure offers a visual method to comprehend charge distribution and bonding.
Understanding the H3O+ Lewis structure starts with recognizing its formation through protonation of water. When H2O gains a proton (H+), it forms H3O+, commonly known as the hydronium ion. This structure is vital in aqueous solutions as it represents the mechanism of acids donating protons.
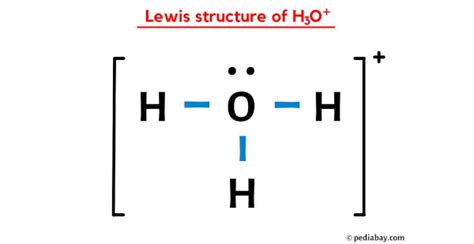
In terms of the molecular geometry, the H3O+ ion exhibits a trigonal pyramidal shape. The central oxygen atom is bonded to three hydrogen atoms, with one of its lone pairs of electrons utilized in each bond. A key aspect of the structure is the positive charge, which resides on the oxygen atom due to its higher electronegativity compared to hydrogen, pulling the shared electrons slightly closer to itself. This creates a polar molecule, essential for its acidic behavior in water.
The importance of valence electrons cannot be overstated when drawing the H3O+ Lewis structure. Oxygen, belonging to Group 16, has six valence electrons, while each hydrogen atom contributes one valence electron. To form the ion, one hydrogen atom donates its electron to oxygen, resulting in a total of eight electrons (the octet rule) around the oxygen, with three hydrogen atoms bonded singly to the oxygen. The remaining lone pair on oxygen accounts for the positive charge (+1).
The H3O+ Lewis structure also emphasizes the role of resonance in explaining the electron delocalization. Although the simplest Lewis structure might suggest a static charge on oxygen, in reality, the proton can shift among the three potential sites, leading to resonance. This dynamic nature of the structure underscores the complexity and behavior of acids in solution.
Analyzing H3O+ within the context of Lewis acids and bases is also enlightening. From a Brønsted-Lowry perspective, H3O+ acts as an acid by donating a proton. According to the Lewis definition, it serves as an electron pair acceptor due to its empty orbital that can accept a pair of electrons from another species, making it a dual acid by both definitions. This dual role emphasizes the importance of understanding both traditional and modern theories of acids and bases in chemistry.
The practical applications of understanding H3O+ extend beyond theoretical chemistry. For example, in biochemical systems, hydronium ions play a crucial role in maintaining the pH of biological fluids. In industrial settings, they are fundamental in processes like pickling and in the manufacture of sulfuric acid through the Contact Process. Recognizing the stability and reactivity of H3O+ helps in optimizing these processes.
What role does H3O+ play in acid-base reactions?
H3O+ acts as a primary acid in aqueous solutions according to the Brønsted-Lowry definition by donating protons. Additionally, it serves as a Lewis acid by accepting electron pairs, showing its dual nature in acid-base chemistry.
How does the H3O+ Lewis structure help in understanding molecular geometry?
The H3O+ Lewis structure illustrates a trigonal pyramidal geometry due to the bonding of three hydrogen atoms with the central oxygen atom, which also holds a positive charge from one of its lone pairs. This visualization helps in comprehending the spatial arrangement of atoms and the distribution of electron density in the molecule.
In conclusion, the H3O+ Lewis structure is not just a simple academic exercise but a crucial element for comprehending the behavior of acids in various scientific and industrial contexts. Through practical insights, evidence-based understanding, and real-world examples, we gain a comprehensive view of this vital chemical species.