Understanding the Unique Traits of Each State of Matter: A Comprehensive Guide
Each state of matter - solid, liquid, gas, and plasma - has its unique set of characteristics and behaviors. Understanding these traits is not only fundamental in scientific education but also immensely practical for everyday tasks. Whether you’re cooking, engineering, or even just observing nature, knowing the properties of these states can enhance your understanding and application. This guide will take you through the unique attributes of each state of matter, providing actionable advice and practical solutions to everyday challenges you may face.
One of the most common problems users face is confusion over the differences between these states. For example, why does ice melt on a hot day, or why does gas expand? This guide aims to clear up these confusions by presenting a step-by-step explanation of each state’s unique traits, supplemented with practical examples and clear, actionable tips.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: To quickly understand the differences, look up basic science textbooks or reputable online sources for visual explanations.
- Essential tip with step-by-step guidance: To identify a state of matter, examine the substance for fixed volume and shape (solid), fixed volume but no fixed shape (liquid), or no fixed volume and shape (gas), and observe under different temperatures.
- Common mistake to avoid with solution: Avoid assuming all liquids behave similarly just because they are both liquids; each liquid has distinct properties and behaviors depending on its composition.
Let’s delve into the details, beginning with the solid state.
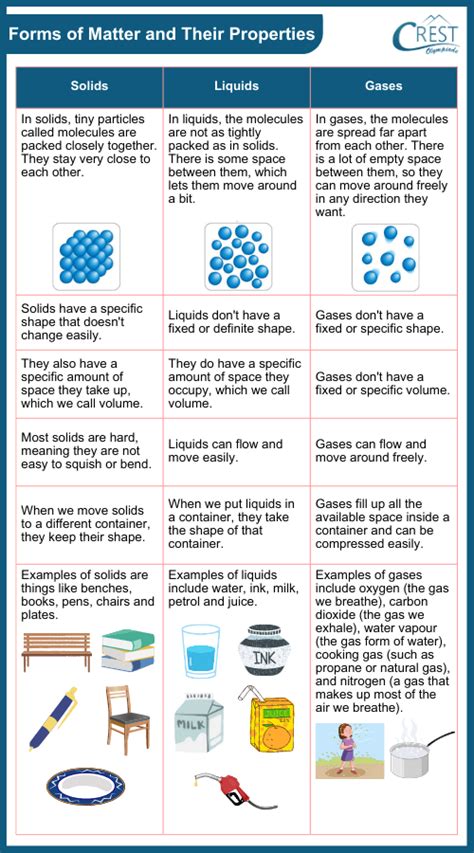
Solid State: Definition and Characteristics
In the solid state, particles are tightly packed together in a fixed structure. This state is defined by a definite volume and shape, which are preserved regardless of the container. This rigidity results from strong intermolecular forces that keep particles aligned and fixed in place.
For instance, a piece of ice retains its shape and volume whether it’s placed in a bowl or a bottle. This trait is useful in various practical applications such as thermal insulation, where solids can be used to protect against heat transfer.
Here’s a step-by-step guide to understanding the solid state more deeply:
- Identify particles: Examine the particles that make up the substance. In solids, particles are closely packed in a regular, repeating pattern.
- Determine shape and volume: A solid has both a fixed shape and a fixed volume.
- Assess intermolecular forces: Strong intermolecular forces hold the particles together, preventing movement.
A practical application of this understanding is in materials science, where engineers use solid materials with specific thermal and mechanical properties for construction and manufacturing.
To avoid common mistakes, remember that while all solids have these traits, the specific characteristics depend on the type of solid. Different solids have varying strengths, melting points, and other properties.
Liquid State: Definition and Characteristics
In the liquid state, particles are close together but not in a fixed structure. Liquids have a definite volume but no fixed shape, taking the shape of their container. The particles in a liquid are close enough to stay together but far enough apart to slide past each other.
Consider water in a glass; it conforms to the shape of the glass while retaining a constant volume, regardless of the container's shape. This property is essential in fluid dynamics, allowing liquids to flow and transfer heat effectively.
Here’s a detailed how-to guide to understanding the liquid state:
- Examine particle arrangement: In a liquid, particles are closely packed but not in a fixed position; they can move around.
- Determine volume and shape: Liquids have a definite volume but take the shape of their container.
- Assess particle movement: Particles can move past each other, which allows the liquid to flow.
Practical examples abound in everyday life, from the way liquids are used in cooking (e.g., pouring oil or water into pans) to their role in transporting nutrients in biological systems.
To avoid common mistakes, remember that while all liquids share these traits, their specific behaviors can vary based on composition, such as viscosity and surface tension. For instance, honey has a higher viscosity than water, affecting how it flows.
Gas State: Definition and Characteristics
In the gas state, particles are far apart and move freely. Gases have neither a fixed volume nor a fixed shape; they expand to fill the entirety of their container. The particles in a gas move rapidly and continuously, colliding with each other and the walls of the container.
Take air in a balloon as an example; the gas fills the balloon’s entire space, adapting to its shape while maintaining the volume that the balloon can hold. This state is critical in processes such as combustion, weather systems, and even respiration.
Here’s a detailed how-to guide to understanding the gas state:
- Examine particle arrangement: Gas particles are widely spaced and move freely in all directions.
- Determine volume and shape: Gases take the shape and volume of their container.
- Assess particle movement: Gas particles move independently, colliding with each other and the container’s walls.
Practical applications include using gases in chemical reactions and understanding atmospheric conditions for weather prediction.
To avoid common mistakes, note that while all gases expand to fill their containers, different gases behave differently based on temperature and pressure. For example, gases expand more under higher temperatures, a principle used in many engineering applications.
Plasma State: Definition and Characteristics
Plasma, the fourth state of matter, consists of ionized gas particles. In plasma, some or all of the electrons are stripped from their atoms, creating a soup of charged particles. This state has neither a fixed volume nor a fixed shape; like gases, plasmas expand to fill their containers. However, plasmas can conduct electricity and respond strongly to magnetic fields.
One of the most remarkable examples is the plasma in neon signs, where a small amount of current ionizes the gas, causing it to emit light. Plasmas also play a crucial role in stars, including the Sun, and in modern technology like plasma screens.
Here’s a detailed how-to guide to understanding the plasma state:
- Examine particle arrangement: Plasma contains ions (charged atoms) and free electrons.
- Determine volume and shape: Plasma, like a gas, takes the shape and volume of its container.
- Assess particle movement: Ions and electrons move freely, conducting electricity and responding to magnetic fields.
Practical examples of plasma usage include applications in electronics, medical procedures like plasma therapy, and various industrial processes.
To avoid common mistakes, remember that although plasmas and gases share traits of expanding to fill their containers, the interactions between charged particles in plasmas differ significantly, making them suitable for specialized applications.
Practical FAQ
How can I distinguish between a solid, liquid, and gas?
To distinguish between these states, observe the shape and volume of the substance:
- Solid: Has a fixed shape and volume.
- Liquid: Has a fixed volume but takes the shape of its container.
- Gas: Neither has a fixed shape nor volume; it will fill the entire container.
Additionally, consider particle movement: in solids, particles are fixed; in liquids, they can move past each other; in gases, they move freely.
What are some common applications of plasma?
Plasma has several practical applications, including:
- Neon signs: Plasma emits light when charged with electricity.
- Medical treatments