The study of molecular geometry and bonding has always held a special place in the realm of chemistry. One compelling topic within this discipline is the exploration of F2 Lewis structure. This analysis delves into the structural arrangement, bonding, and characteristics of molecular fluorine. Understanding the intricacies of F2 Lewis structure provides valuable insights for both novice and seasoned chemists, opening doors to comprehending molecular interactions and reactivity.
Key Insights
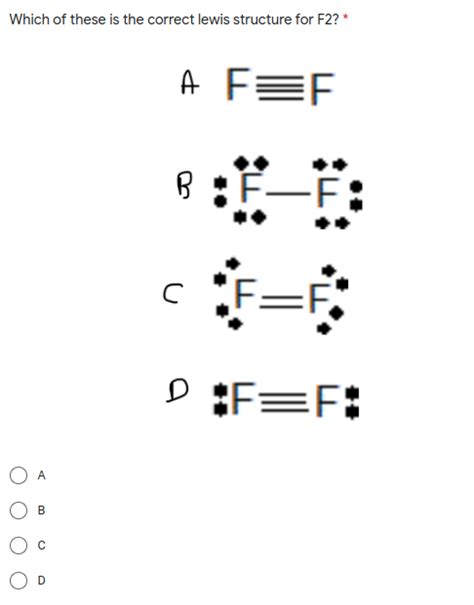
- F2 molecule consists of two fluorine atoms sharing a single bond
- The Lewis structure reveals the absence of lone pairs on the fluorine atoms
- Simple yet crucial, understanding F2 helps in grasping molecular bonding basics
Understanding the Basics of F2 Lewis Structure
F2 Lewis structure, at its core, represents a diatomic molecule of fluorine. Each fluorine atom has seven valence electrons, and the molecule forms a single covalent bond to achieve stability. This bond is formed by the sharing of one pair of electrons between the two fluorine atoms. The simplicity of this structure allows chemists to easily extrapolate to more complex molecules, understanding the fundamental concept of electron sharing in covalent bonds.Electron Configuration and Bond Formation
To analyze F2’s Lewis structure in detail, we delve into the electron configuration. The fluorine atom, positioned at atomic number seven, exhibits an electron configuration of 1s² 2s² 2p⁵. For the F2 molecule, both atoms share one electron, filling their respective valence shells. Consequently, each fluorine atom attains a full outer shell similar to the noble gas neon, thus achieving octet stability. The single bond formed between the two fluorine atoms is a clear illustration of how atoms achieve electronic stability through sharing.The Implications for Chemical Reactivity
Understanding the F2 Lewis structure has practical implications for chemical reactivity and molecular behavior. Fluorine, being highly reactive, forms strong bonds due to its small atomic radius and high electronegativity. The simplicity of the F2 molecule helps to illustrate the reactivity of fluorine and its tendency to gain or share electrons to form bonds with other elements. This fundamental knowledge is essential for predicting and understanding the chemical behavior of fluorine in various reactions, including its role in organic and inorganic synthesis.How does F2 bonding differ from other diatomic molecules?
While many diatomic molecules like H2 or N2 form similar covalent bonds, F2’s high electronegativity results in a stronger bond compared to less electronegative elements. This distinction is crucial for understanding the reactivity and bond strength in different diatomic molecules.
Can the F2 molecule form multiple bonds?
F2, due to its high electronegativity and the nature of its valence electrons, typically forms single bonds. It does not typically form double or triple bonds, which are more common in less electronegative elements like carbon or nitrogen.