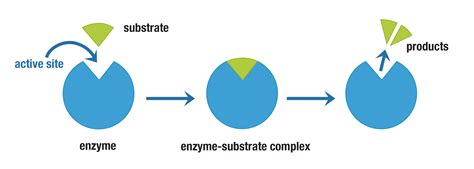
In the world of biochemistry, enzyme substrate complex formation is a fundamental concept that drives countless biological processes. Many users often struggle with understanding how enzymes interact with their substrates, forming complexes that ultimately lead to the catalysis of various biochemical reactions. This guide is designed to walk you through the intricate dance between enzymes and substrates, offering practical steps to help you grasp this crucial biochemical phenomenon.
Imagine you’re trying to unlock a door to a hidden treasure chest. The key is your enzyme, the lock is your substrate, and the combination to make them work together is the enzyme-substrate complex. By understanding the steps to forming this complex, you'll be able to open this treasure chest of biological reactions. Let's dive into how to achieve this with practical, actionable advice.
Getting Started: The Basics of Enzyme-Substrate Complex Formation
The formation of an enzyme-substrate complex is like a precise handshake between an enzyme and a substrate. This process is central to understanding enzyme kinetics and the regulation of metabolic pathways. Without this understanding, you’ll miss out on the subtleties that control your biological systems. Here’s how you can start:
Quick Reference
- Immediate action item with clear benefit: Understand the active site of the enzyme where the substrate binds. This will enable you to see the specific interaction sites.
- Essential tip with step-by-step guidance: Draw the enzyme and substrate, then mark the active site on the enzyme where the substrate fits precisely like a key into a lock.
- Common mistake to avoid with solution: Overlooking the importance of enzyme specificity; each enzyme has a unique active site that can only bind to a specific substrate. Use molecular modeling tools to simulate the interaction and confirm specificity.
By grasping these basics, you'll be equipped to delve deeper into the nuanced interactions governing enzyme-substrate complex formation.
Step-by-Step Guide to Forming Enzyme-Substrate Complexes
To fully understand enzyme-substrate complex formation, let’s walk through each step in a methodical and practical way:
Step 1: Identifying the Enzyme’s Active Site
The enzyme's active site is where the magic happens. This specific region on the enzyme provides a snug fit for the substrate. It’s shaped to match the substrate like a mold. To identify this:
- Consult the enzyme’s structure through crystallographic data or molecular modeling.
- Look for the region where binding to the substrate will induce a conformational change, often referred to as the "induced fit."
By identifying the active site, you get a clear picture of where the substrate will bind, leading to the formation of the complex.
Step 2: Preparing the Substrate
Just like a key needs to be ready to fit into a lock, your substrate must be in the proper condition to bind with the enzyme. Preparation steps can include:
- Ensuring the substrate is at the right concentration.
- Confirming the substrate is in the correct chemical form and charge state for binding.
If the substrate needs any modification before binding, apply those modifications. For instance, some substrates need to be dephosphorylated or oxidized to bind.
Step 3: The Lock and Key Mechanism
The initial interaction between the enzyme and substrate is like a key entering a lock. Here’s what happens during this interaction:
- The substrate approaches the active site of the enzyme.
- Specific binding interactions (hydrogen bonds, ionic bonds, etc.) form between the substrate and the active site.
- The enzyme induces a fit, changing shape to better fit the substrate, enhancing the stability of the complex.
At this stage, the enzyme-substrate complex is formed. This interaction is crucial and sets the stage for catalysis.
Step 4: Catalytic Action and Product Formation
With the complex formed, the enzyme lowers the activation energy of the reaction. This is where the catalytic action occurs, transforming the substrate into the product. Follow these steps:
- The enzyme facilitates the chemical reaction by providing an alternative reaction pathway with a lower activation energy.
- The substrate is converted into the product within the active site.
- Once the reaction is complete, the product is released, and the enzyme returns to its original state, ready to bind another substrate.
Understanding each step ensures you can see the full cycle of enzyme action from binding to product release.
Advanced Tips and Tricks
Once you’ve mastered the basics, you’ll want to delve into more advanced concepts to refine your understanding:
- Study allosteric regulation, where effectors bind to an enzyme away from the active site, changing its functionality.
- Investigate competitive and non-competitive inhibition, understanding how inhibitors can block enzyme action.
- Look into enzyme kinetics, focusing on parameters like Km and Vmax, which provide deeper insights into enzyme efficiency and substrate binding.
These advanced topics allow you to appreciate the complex control mechanisms enzymes exert over biochemical pathways.
Practical FAQ
What common mistakes should I avoid when studying enzyme-substrate complex formation?
Avoid the following common pitfalls:
- Misidentifying the active site: Always double-check the enzyme's 3D structure to ensure you’re looking at the correct binding site.
- Overlooking enzyme specificity: Remember each enzyme has unique specificity. Mistaking one enzyme for another can lead to incorrect assumptions.
- Ignoring experimental conditions: pH, temperature, and ionic strength can greatly affect enzyme activity and complex formation.
By keeping these points in mind, you’ll minimize errors and build a robust understanding of enzyme-substrate interactions.
Through this detailed guide, you now have a comprehensive understanding of enzyme substrate complex formation. From identifying active sites to understanding catalytic actions, you’re equipped with practical steps and tips to master this essential biochemical process.