P
Understanding the nuances of chemical bonding is critical for advancements in chemistry, materials science, and related fields. A key concept is the type of bond in which electrons are transferred, a process integral to many chemical reactions, from ionic compounds to electrochemical cells. This article aims to delve into this mechanism, providing practical insights into its implications, applications, and the fundamental principles underlying electron transfer.
Key Insights
- Primary insight with practical relevance: Electron transfer is fundamental in understanding ionic bond formation and plays a crucial role in processes such as corrosion and battery function.
- Technical consideration with clear application: The ability to predict electron transfer in a given compound can aid in the design of new materials with specific electrical or structural properties.
- Actionable recommendation: Emphasizing electron transfer dynamics in educational curricula can foster a deeper understanding of chemical interactions.
Electron Transfer in Ionic Bonds
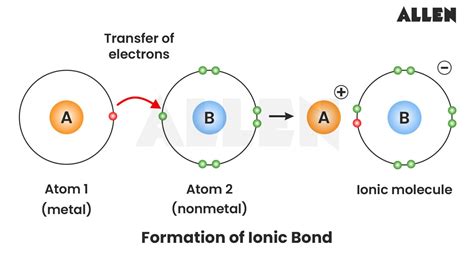
In the realm of ionic bonds, the transfer of electrons from one atom to another is the cornerstone. This electron donation from a metal to a non-metal results in the formation of ions: a positively charged cation and a negatively charged anion. A classic example is the formation of sodium chloride (NaCl). Here, a sodium atom (Na) loses an electron to become Na+, while a chlorine atom (Cl) gains an electron to become Cl-, resulting in an ionic bond between Na+ and Cl-. This electron transfer is not merely a theoretical curiosity; it underpins the structure and properties of many compounds used in everyday life, from table salt to industrial ceramics.
Electron Transfer in Electrochemical Applications
Beyond simple ionic bonding, electron transfer is pivotal in electrochemical processes, which are the bedrock of many modern technologies. For instance, in batteries, electron transfer between the anode and cathode drives the flow of electric current. In a lithium-ion battery, lithium ions migrate from the cathode to the anode during discharge, with electrons following the external circuit. This process is contingent on efficient electron transfer mechanisms, which can significantly influence the battery’s performance, charge/discharge rates, and overall lifespan. Understanding these principles allows chemists and engineers to design better batteries with higher energy densities and longer operational cycles.
What role does electron transfer play in corrosion?
Electron transfer is a key factor in the process of corrosion, particularly in metals. The natural tendency of a metal to lose electrons to more electronegative elements or compounds leads to the degradation of the metal, forming oxides or other compounds. Understanding this process helps in developing protective coatings and strategies to mitigate corrosion in critical applications.
How can understanding electron transfer improve material design?
By comprehending electron transfer dynamics, scientists can tailor the electronic properties of materials for specific applications. For example, materials designed for semiconductor devices require precise control over electron movement to achieve desired electrical characteristics. This understanding allows for the creation of materials with enhanced conductivity, improved stability, or other tailored properties.
This exploration underscores the significance of electron transfer in chemical bonding and technology. From the straightforward formation of ionic compounds to the intricate workings of advanced electrochemical devices, understanding this process enables progress across various scientific domains. Emphasizing this concept not only advances theoretical knowledge but also fosters innovation in practical applications, illustrating the profound impact of fundamental chemical principles on modern technology and everyday life.