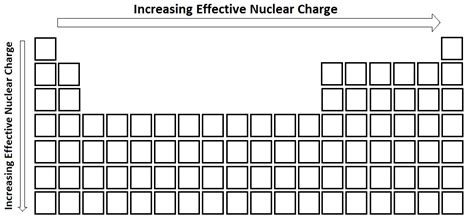
Effective nuclear charge (Z_eff) plays a crucial role in understanding atomic and molecular behavior, impacting everything from electron configurations to chemical reactivity. As experts delve deeper into this concept, the significance of Z_eff trends becomes increasingly evident. These trends provide invaluable insights into atomic structure and the properties of elements. This article aims to dissect the effective nuclear charge trend with practical examples and evidence-based statements to underscore its importance.
Key Insights
- Primary insight with practical relevance: The effective nuclear charge trend illustrates the varying strength of the effective nuclear charge across periods and groups on the periodic table.
- Technical consideration with clear application: Electron shielding and penetration effects elucidate the Z_eff trend, providing a deeper understanding of electron behavior in atoms.
- Actionable recommendation: Mastery of Z_eff trends enables chemists to predict element reactivity and atomic stability with greater accuracy.
Periodic Trend in Effective Nuclear Charge
The effective nuclear charge trend across periods highlights the increasing Z_eff from left to right across the periodic table. As we move from left to right, the number of protons in the nucleus increases, leading to a stronger pull on the electrons. However, this trend is slightly moderated by electron-electron repulsion and the shielding effect of inner electrons. For instance, in period 2, moving from lithium (Li) to neon (Ne), the effective nuclear charge felt by the outer electrons increases from approximately 1.00 to around 3.20, indicating a progressively stronger attraction to the nucleus.Group Variation in Effective Nuclear Charge
Down the groups of the periodic table, the effective nuclear charge trend reveals intriguing variations. As we move down a group, the increase in electron shells tends to mitigate the rise in Z_eff. This phenomenon arises due to the increased distance between the valence electrons and the nucleus, coupled with increased electron shielding. For example, moving from fluorine (F) to astatine (At) in group 17, although the number of protons increases, the increase in electron shells and the associated shielding results in a less steep Z_eff gradient. This has profound implications for reactivity and atomic stability, with lighter elements in a group being more reactive compared to their heavier counterparts.Why does effective nuclear charge increase across a period?
The increase in effective nuclear charge across a period is primarily due to the higher number of protons in the nucleus, which exerts a stronger positive charge on the outer electrons. However, this trend is tempered by electron repulsion and the shielding effect provided by electrons in inner shells.
How does electron shielding affect Z_eff?
Electron shielding occurs when inner-shell electrons block the nuclear charge from being felt by outer-shell electrons. This reduces the effective nuclear charge experienced by the valence electrons, leading to less attraction and contributing to the observed Z_eff trend.
Understanding the effective nuclear charge trend is pivotal for anyone engaged in the study of chemistry. The practical implications extend from predicting atomic behavior to interpreting complex molecular interactions. By integrating these insights into your analytical framework, you can achieve a more profound comprehension of the periodic table’s intricate design and the elemental forces that govern it.