Understanding Coordinate Covalent Compounds: A Practical User-Focused Guide
Coordinate covalent compounds, also known as coordination compounds, are fascinating molecules where central metal ions are surrounded by molecules or anions that act as ligands. These compounds play an essential role in various biological and industrial applications. Whether you’re a student delving into chemistry for the first time or a professional looking to deepen your knowledge, this guide aims to demystify coordinate covalent compounds with clear, actionable advice and real-world examples.
Coordinate covalent compounds can often seem complex, but with a step-by-step approach, you'll find them easier to grasp. Many students and professionals struggle to understand how these compounds form, their structural configurations, and their practical applications. This guide will walk you through the fundamental concepts, practical examples, and problem-solving strategies to ensure you have a solid understanding of coordinate covalent compounds.
Quick Reference
Quick Reference
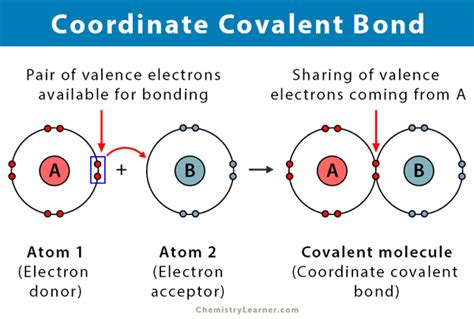
- Immediate action item with clear benefit: Always remember that the central metal ion provides empty orbitals while the ligands provide lone pairs of electrons for bond formation.
- Essential tip with step-by-step guidance: To determine if a compound is a coordinate covalent compound, identify if there is a metal ion and ligands donating a pair of electrons to the metal.
- Common mistake to avoid with solution: Avoid confusing coordinate covalent bonds with other types of covalent bonds. Ensure you note that only ligands with lone pairs can form these bonds with the metal ion.
By keeping these points in mind, you'll be better equipped to identify and understand the formation and function of coordinate covalent compounds.
Understanding the Basics of Coordinate Covalent Compounds
To start, let’s break down what makes coordinate covalent compounds unique. At their core, these compounds form when a central metal atom or ion is bonded to ligands, which are molecules or ions that contain at least one lone pair of electrons. This type of bonding is different from traditional covalent bonding, where both electrons in the bond are shared equally between atoms.
A classic example is the hexaaquacopper(II) ion, [Cu(H2O)6]2+. In this compound, the central copper(II) ion has six water molecules (ligands) surrounding it, each donating a lone pair of electrons from oxygen to the copper ion.
To better understand this, follow these steps:
- Identify the central metal atom or ion: In the hexaaquacopper(II) ion, copper (Cu) is the central metal.
- Determine the ligands: The ligands here are the water molecules (H2O).
- Analyze the bonding: Each water molecule donates a lone pair of electrons to the copper ion, forming a coordinate covalent bond.
Through this process, you’ll gain a foundational understanding of the fundamental interactions and structures within coordinate covalent compounds.
Deep Dive into the Formation and Structure
Creating a detailed understanding of coordinate covalent compounds starts with grasping the intricacies of their formation and structural arrangements. The central metal ion’s empty orbitals and the ligands’ lone pairs create a dynamic interaction, leading to stable compounds.
Here’s how the structure forms:
- Central Metal Ion: The central atom typically has vacant d-orbitals available for bonding.
- Ligands: These are electron-rich species that donate a pair of electrons to the metal ion.
- Bonding: When a ligand donates a lone pair to the central metal ion, a coordinate covalent bond is formed.
Consider another example, hexacarbonyliron(0), Fe(CO)6. In this compound, iron has six carbonyl (CO) ligands surrounding it. Each CO ligand donates a lone pair of electrons from the oxygen atom to the iron atom.
To identify the structure:
- Identify the central atom: Iron (Fe).
- Identify the ligands: Carbon monoxide (CO).
- Determine the coordination number: The iron atom is surrounded by six CO ligands, indicating a coordination number of six.
- Understand the geometry: This arrangement leads to an octahedral geometry around the central iron atom.
This systematic approach ensures that you can accurately determine the structure and configuration of any coordinate covalent compound.
Real-World Applications
Coordinate covalent compounds are more than theoretical constructs—they have practical applications in numerous fields. For example, chlorophyll, the green pigment in plants that captures light for photosynthesis, is a coordination compound containing magnesium at its center.
Additionally, many industrial catalysts are coordination compounds. One significant example is ferrocene, Fe(C5H5)2, which has applications in organic synthesis and serves as a prototype for studying organometallic chemistry.
Understanding the real-world applications will emphasize the importance of studying these compounds and prepare you for their use in various sectors.
Practical FAQ
What are the common uses of coordinate covalent compounds in industry?
Coordinate covalent compounds find extensive uses in industry, particularly in catalysis and as pigments. Industrial catalysts, which speed up chemical reactions without being consumed in the process, often utilize coordination compounds. For example, platinum group metals in coordination compounds are used in catalytic converters to reduce harmful emissions in vehicle exhaust. Coordination compounds also play a critical role in the production of dyes and pigments, such as Prussian blue, used in paints and inks.
The practical uses in industry highlight how understanding these compounds can lead to innovations and improvements in various sectors.
Coordinate covalent compounds might initially appear intricate, but with this guide, you’ve taken a significant step towards understanding their formation, structure, and applications. By following the step-by-step guidance provided, you’ll be well-equipped to recognize and explore the fascinating world of coordination chemistry.