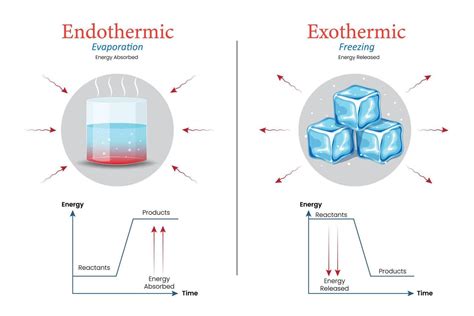
The process of condensation is fundamental in various scientific and industrial applications, raising the question: Is condensation exothermic or endothermic? Understanding this concept not only aids in theoretical comprehension but also holds significant practical implications in fields ranging from meteorology to HVAC systems.
The crux of condensation lies in its thermal characteristics. Condensation occurs when water vapor transitions from a gaseous state to a liquid state, releasing latent heat in the process. This makes it an exothermic process. Essentially, as water vapor cools and condenses, it loses energy in the form of heat, transferring this energy to its surroundings. The heat released during condensation helps to maintain temperature stability in various environments.
Key Insights
- Primary insight with practical relevance: Condensation is an exothermic process. Understanding this concept can help in designing efficient cooling systems and better understanding weather patterns.
- Technical consideration with clear application: Recognizing that condensation releases heat is crucial for optimizing HVAC systems to enhance energy efficiency and reduce utility costs.
- Actionable recommendation: Utilize the exothermic nature of condensation in passive cooling strategies to improve building thermal management.
Scientific Underpinning of Condensation
Scientifically, the transition from vapor to liquid during condensation involves a phase change. This phase change is inherently accompanied by the release of latent heat, which is energy required for the phase transition from gas to liquid without altering temperature. According to thermodynamic principles, exothermic reactions release energy in the form of heat. Thus, condensation aligns perfectly with these principles as it releases latent heat, confirming its exothermic nature. To grasp this better, consider the formation of dew on a cool surface at night; the latent heat released during this condensation process is what warms the surface slightly.
Practical Applications of Condensation
The exothermic nature of condensation carries significant practical implications across various fields. In HVAC systems, for instance, understanding condensation can lead to better control of indoor climate. By exploiting the heat released during condensation, systems can be designed to use this released energy to warm surrounding areas passively. Additionally, in large industrial setups, condensation can be harnessed to preheat incoming cold water, minimizing energy use. Furthermore, meteorological studies benefit from this knowledge to predict weather patterns, especially in the formation of clouds where condensation plays a pivotal role.
Does the exothermic nature of condensation mean that it can be harnessed for cooling?
While condensation is exothermic, it releases heat rather than absorbing it for cooling. However, understanding the heat released can help in designing systems where this heat can be reused or managed effectively to optimize energy efficiency.
Why is it important to understand whether condensation is exothermic or endothermic?
Understanding whether condensation is exothermic or endothermic is crucial for designing effective thermal management systems, enhancing energy efficiency, and making informed decisions in various engineering and environmental applications.
This exploration reveals that condensation is unequivocally an exothermic process, a fact that holds substantial practical value in engineering and environmental sciences. With this knowledge, we can innovate and optimize systems for improved energy use and environmental management.